Biologic Product Portfolio — Educational & Regulatory Overview
Not By Chance Life LLC facilitates credential-based documentation access and structured supplier evaluation for biologic product categories reviewed within the 21 CFR Part 1271 Section 361 regulatory framework.
Regulatory classification considerations may involve evaluation under the Section 361 vs Section 351 HCT/P regulatory distinction, including analysis of the minimal manipulation regulatory standard and homologous use regulatory criteria, as well as compliance with the 21 CFR Part 1271 Subparts A–D regulatory structure and Current Good Tissue Practice (cGTP) regulatory safeguards.
Regulatory positioning considerations and enforcement context are further discussed within the FDA enforcement landscape for HCT/P regulation.
Manufacturer processing, donor screening, laboratory validation, and fulfillment activities are conducted directly by Regenerative Research Group (RRG) in accordance with establishment registration and current good tissue practice standards under 21 CFR Part 1271.
Biologic Product Education Framework
The following educational reference illustrates biologic category groupings, sourcing transparency standards, regulatory classification considerations, and documentation traceability elements reviewed during supplier evaluation.
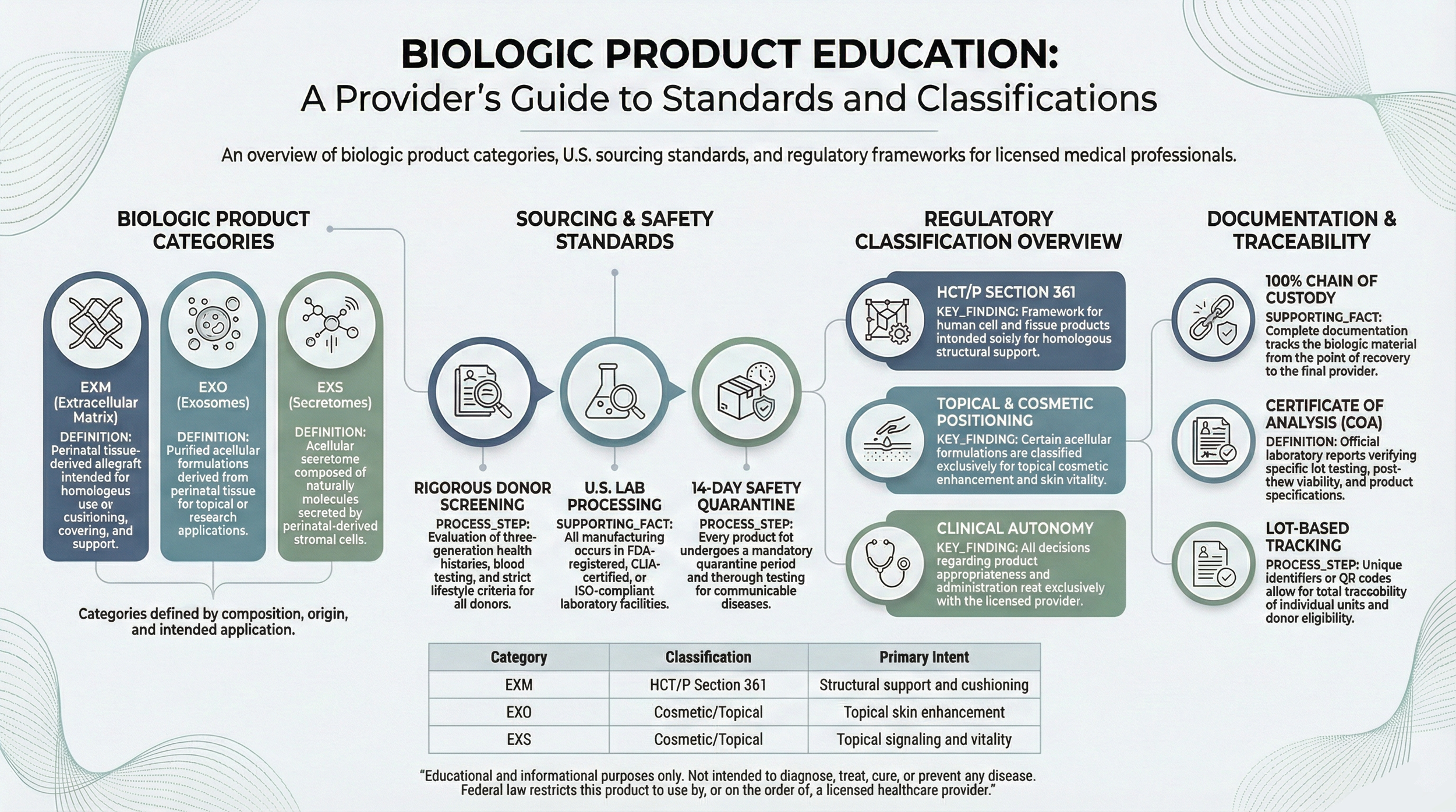
Structured Supplier Evaluation Process
Licensed professionals evaluating biologic categories may review the structured supplier evaluation and documentation transparency process prior to credential verification.
Extracellular Matrix (EXM) Allograft Overview
Structural tissue-derived allograft material presented for professional documentation review within the HCT/P regulatory classification structure.
Exosome-Related Formulations Overview
Composition-focused extracellular vesicle-associated formulations presented for regulatory classification analysis and documentation transparency review.
Secretome Formulation Overview
Non-cellular biologic formulations containing soluble molecular components presented for professional regulatory evaluation and sourcing documentation review.
Professional Documentation Access
Access to manufacturer documentation, specifications, sourcing transparency records, and lot-level traceability materials is restricted to verified licensed healthcare professionals.
This information is provided for educational purposes only. Certain materials discussed may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under the 21 CFR Part 1271 Section 361 regulatory framework. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, endorsement, or evaluation of safety or effectiveness. This information is not intended to diagnose, treat, cure, or prevent any disease.
Submission does not guarantee approval. Credentials are manually reviewed prior to granting access to supplier documentation or distribution coordination.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361.
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

