Peer-Reviewed Scientific References
The following peer-reviewed publications provide general scientific background relevant to extracellular matrix biology, placental tissue structure, extracellular vesicle research, secretome biology, and the federal regulatory framework governing 21 CFR Part 1271 Section 361 .
These references are provided for educational context only and do not constitute product-specific claims, clinical validation, or regulatory endorsement.
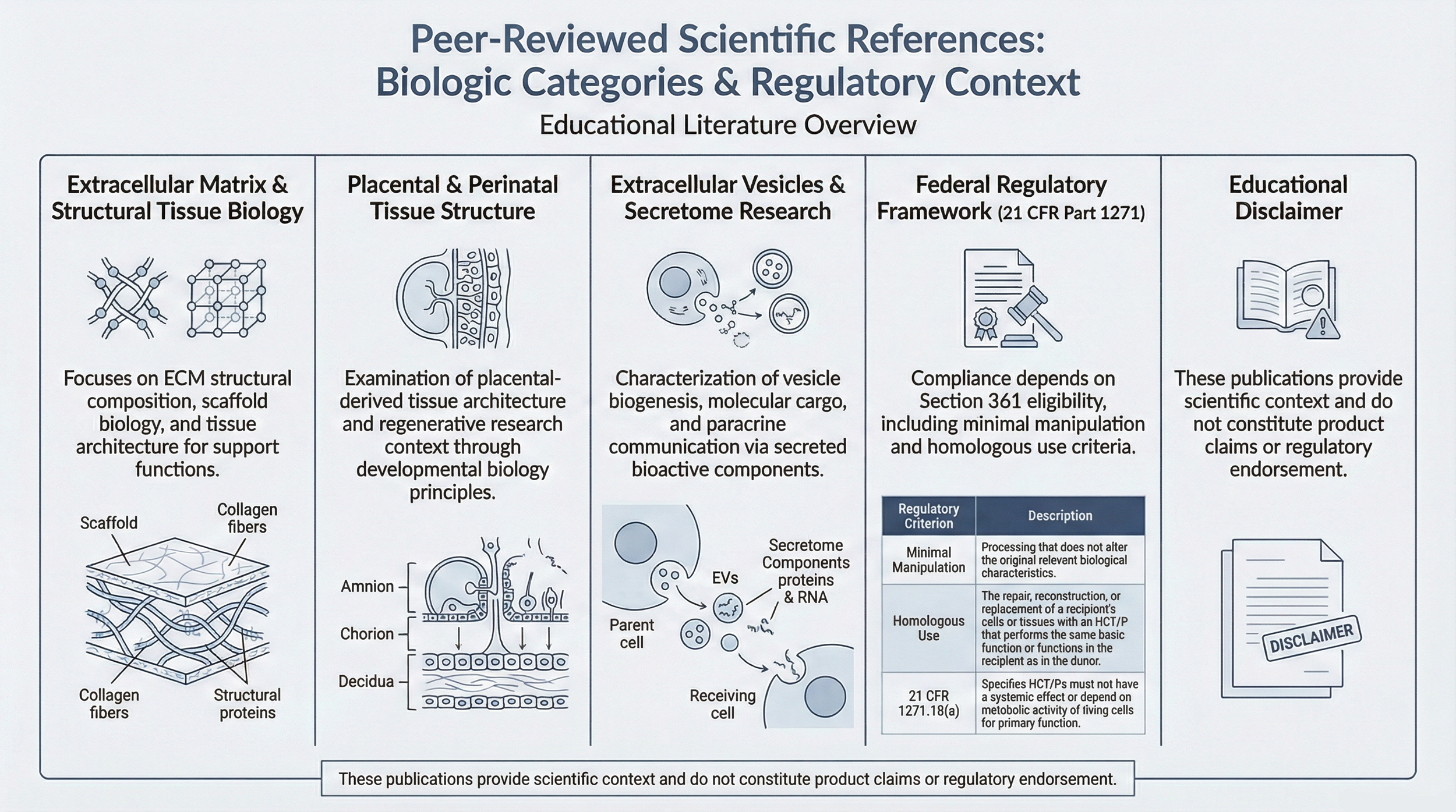
Extracellular Matrix & Structural Tissue Biology
Frantz C, Stewart KM, Weaver VM. The extracellular matrix at a glance. Journal of Cell Science. 2010;123(Pt 24):4195–4200.
Hynes RO. The extracellular matrix: not just pretty fibrils. Science. 2009;326(5957):1216–1219.
Badylak SF. The extracellular matrix as a biologic scaffold material. Biomaterials. 2007;28(25):3587–3593.
Placental & Perinatal Tissue Structure
Parolini O, et al. Toward cell therapy using placental-derived cells: disease mechanisms, cell biology, preclinical studies, and regulatory aspects. Stem Cells and Development. 2010;19(2):143–154.
Silini AR, et al. The long path of human placenta in regenerative medicine. Frontiers in Bioengineering and Biotechnology. 2015;3:162.
Extracellular Vesicles & Exosome Biology
Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis, and function. Nature Reviews Immunology. 2002;2(8):569–579.
Yáñez-Mó M, et al. Biological properties of extracellular vesicles. Journal of Extracellular Vesicles. 2015;4:27066.
Kalluri R, LeBleu VS. The biology and biomedical applications of exosomes. Science. 2020;367(6478).
Extracellular vesicle and exosome research represents an active area of biological investigation. Products derived from extracellular vesicles may be regulated as biological drugs requiring appropriate FDA authorization pathways.
Secretome & Cell Signaling Research
Vizoso FJ, et al. Mesenchymal stem cell secretome: toward cell-free therapeutic strategies. International Journal of Molecular Sciences. 2017;18(9):1852.
Ferreira JR, et al. Mesenchymal stromal cell secretome: influencing therapeutic potential. Journal of Cellular Physiology. 2018;233(5):3731–3744.
Publications referenced in this section describe experimental cell-derived signaling environments and do not represent approved clinical applications or regulatory classification for human tissue products.
Federal Regulatory Framework
U.S. Food and Drug Administration. 21 CFR Part 1271 – Human Cells, Tissues, and Cellular and Tissue-Based Products .
FDA Guidance: Minimal Manipulation and Homologous Use considerations under the Public Health Service Act.
Additional structured explanations are available within:
Related Regulatory & Evaluation Resources
Educational reference only. Inclusion of these publications does not imply FDA approval, clearance, evaluation of safety or effectiveness, or endorsement of any specific product. Certain materials referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361.

