Extracellular Matrix (EXM) Allograft Overview
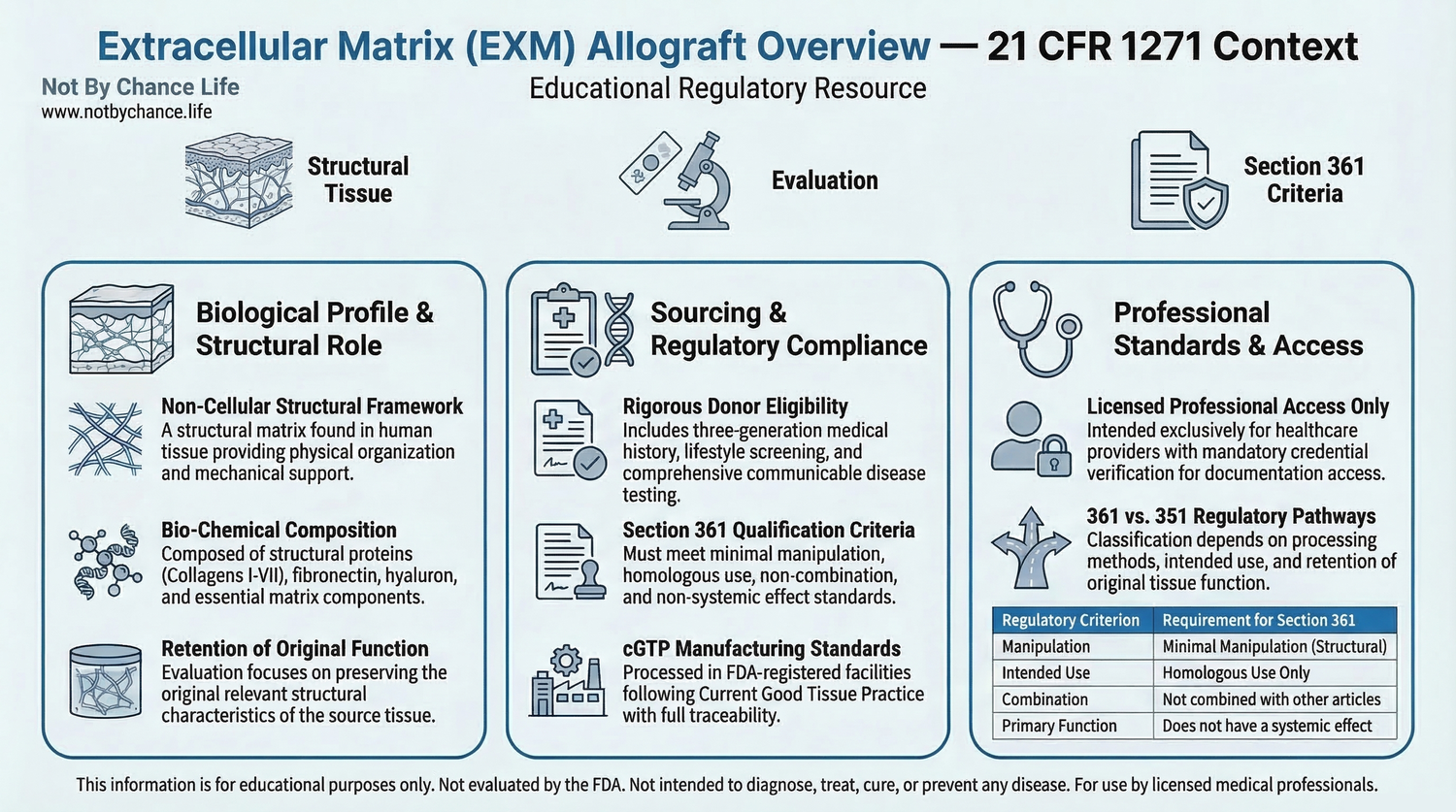
Educational infographic outlining extracellular matrix (EXM) allograft characteristics and regulatory evaluation considerations under 21 CFR Part 1271.
Extracellular Matrix (EXM) refers to the structural, non-cellular framework naturally present within human tissues. In certain biologic contexts, extracellular matrix material may be derived from placental or umbilical tissue sources and processed in accordance with federal tissue regulations.
This page provides an educational overview of extracellular matrix allograft materials, including structural characteristics, sourcing considerations, and regulatory positioning under 21 CFR Part 1271 Section 361.
This overview is part of the broader Biologic Product Portfolio .
Structural Role of the Extracellular Matrix
The extracellular matrix provides physical structure and support to surrounding cells within native tissue. It consists of structural proteins and other non-cellular components that contribute to the tissue’s mechanical and organizational properties.
When derived and processed in compliance with regulatory standards, extracellular matrix materials may be categorized as structural tissue allografts.
Non-Cellular Characteristics
Extracellular matrix allografts referenced in regulated tissue contexts are typically processed to remove cellular components, resulting in a non-cellular structural material.
Regulatory evaluation often considers whether processing preserves the original relevant structural characteristics in accordance with minimal manipulation standards.
Sourcing & Donor Screening Standards
Placental and umbilical tissue sources used for extracellular matrix materials are subject to donor eligibility determination, communicable disease screening, and laboratory testing requirements under 21 CFR Part 1271 Subparts A–D.
Establishments involved in recovery and processing must comply with current good tissue practice (cGTP) requirements, documentation protocols, and traceability standards.
Regulatory Considerations
Certain extracellular matrix materials may qualify for regulation solely under 21 CFR Part 1271 Section 361 if they meet all applicable criteria, including minimal manipulation, homologous use, and systemic effect limitations.
Classification analysis may also reference the Section 361 vs Section 351 distinction.
Classification depends on processing methods, intended use, documentation, and regulatory interpretation.
Structural Tissue Classification
Because extracellular matrix materials serve a structural function, regulatory review may evaluate whether the material retains its original structural properties after processing.
Additional explanation regarding classification pathways is available in the HCT/P Classification Overview.
Professional Evaluation Pathway
Licensed healthcare professionals seeking documentation transparency, sourcing standards, and regulatory positioning clarification may review the structured Structured Supplier Evaluation Process.
Credential confirmation is required prior to documentation release and manufacturer discussion coordination.
Related Biologic Categories
Reference Materials
For federal regulatory reference, consult:
Educational reference only. Certain products may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease. Federal law restricts distribution to licensed medical professionals or by their order.

