Professional & Institutional Inquiries
Not By Chance Life LLC supports licensed medical professionals, institutional operators, and qualified research entities through structured regulatory education and credential-based documentation access coordination within the 21 CFR Part 1271 Section 361 framework.
For a complete regulatory structure overview, review the Regulatory Framework Overview, including classification criteria such as minimal manipulation and homologous use.
This page is intended exclusively for professional and institutional inquiries. Consumer inquiries are not supported.
Before Submitting an Inquiry
Professionals seeking documentation transparency or regulatory evaluation clarification should review the Structured Supplier Evaluation Process, including considerations such as donor eligibility, current good tissue practices (cGTP), and chain-of-custody documentation standards.
Additional scientific context is available within our Peer-Reviewed Scientific References.
Provider Access Requests
Licensed healthcare providers seeking access to documentation must initiate credential verification through the Provider Credential Verification Portal.
Credential review is conducted manually prior to documentation release. Access is not granted via general email inquiry.
Credential-Based Access Standard
Access to documentation, educational materials, and regulatory references is limited to licensed medical professionals and qualified institutional entities.
Submitted inquiries may be subject to credential verification prior to response.
Requests that do not include sufficient professional or institutional context may not be processed.
Professional Access & Communication Standards
The following visual provides an educational overview of credential-based access and structured communication boundaries.
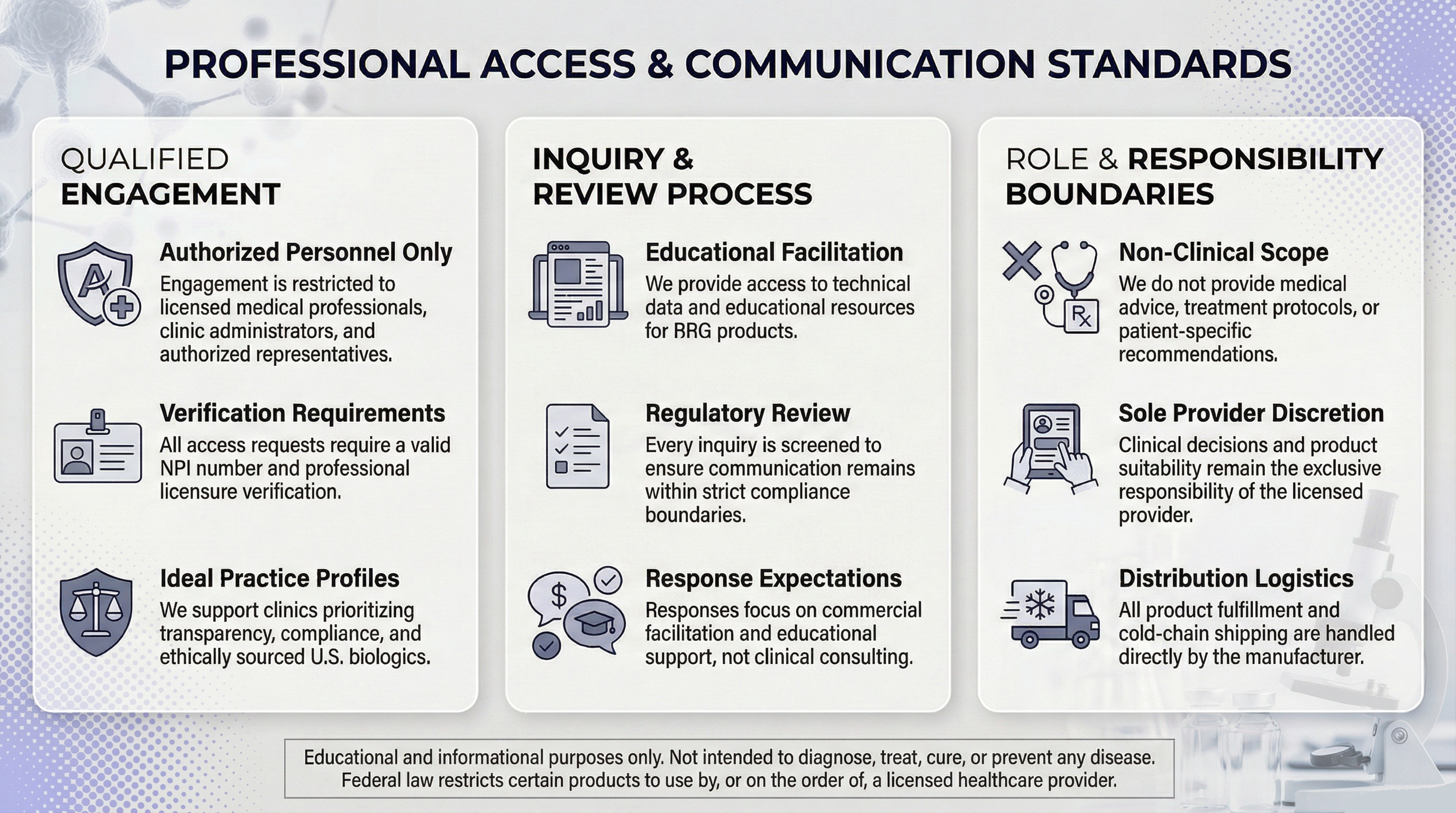
This visual is provided for educational purposes only and does not represent product specifications, clinical use, or treatment frameworks.
Appropriate Institutional Inquiry Categories
- Regulatory framework clarification
- Institutional regulatory alignment discussions
- Multi-location governance coordination
- Documentation and traceability review alignment
- Professional partnership discussions
Professional Communication Channel
Institutional inquiries may be directed to: info@notbychance.life
- Full name
- Professional designation
- Institution affiliation
- Nature of inquiry
Credential verification requests must be submitted via the Provider Portal.
Regulatory & Compliance Notice
This information is for educational purposes only. Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease. For use by licensed medical professionals only.

