Section 361 vs Section 351 HCT/P Classification — Regulatory Distinction
This page explains how Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) are classified under U.S. regulatory frameworks.
This classification is part of the broader regulatory structure defined under 21 CFR Part 1271.
For a complete set of regulatory resources and supporting criteria, see the Regulatory Resource Library.
Regulatory classification determines whether a product may be regulated solely under Section 361 (focused on communicable disease control) or requires regulation as a biological product under Section 351.
Documentation and Regulatory Education
Licensed medical professionals and organizations may request educational materials related to regulatory classification, donor eligibility standards, and documentation practices referenced within this resource.
Credential verification may be required prior to documentation access.
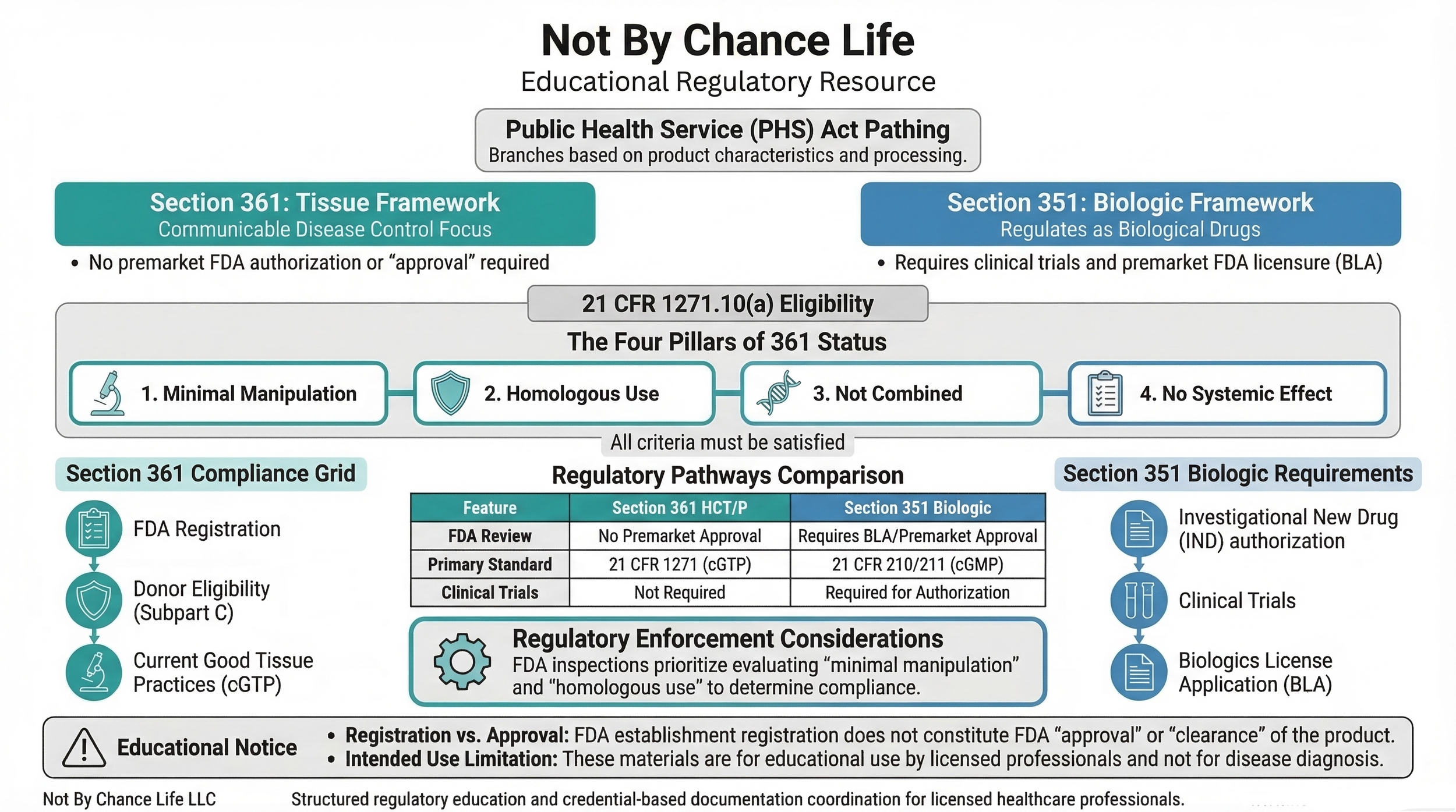
Section 361 Qualification Criteria
- Minimal manipulation
- Homologous use
- Structural vs Cellular Tissue
- Not combined with another article (with limited exceptions)
- No systemic effect (unless qualifying exception applies)
Additional requirements include donor eligibility determination and compliance with Donor Eligibility and Current Good Tissue Practice (cGTP).
Section 351 Regulatory Pathway
- Investigational New Drug (IND) authorization
- Clinical trials
- Biologics License Application (BLA)
- Premarket regulatory review
Products regulated under Section 351 are subject to biologics regulatory pathways and are not eligible for regulation solely under Section 361.
Related Regulatory Topics
Regulatory Notice
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

