Secretome Formulation Overview
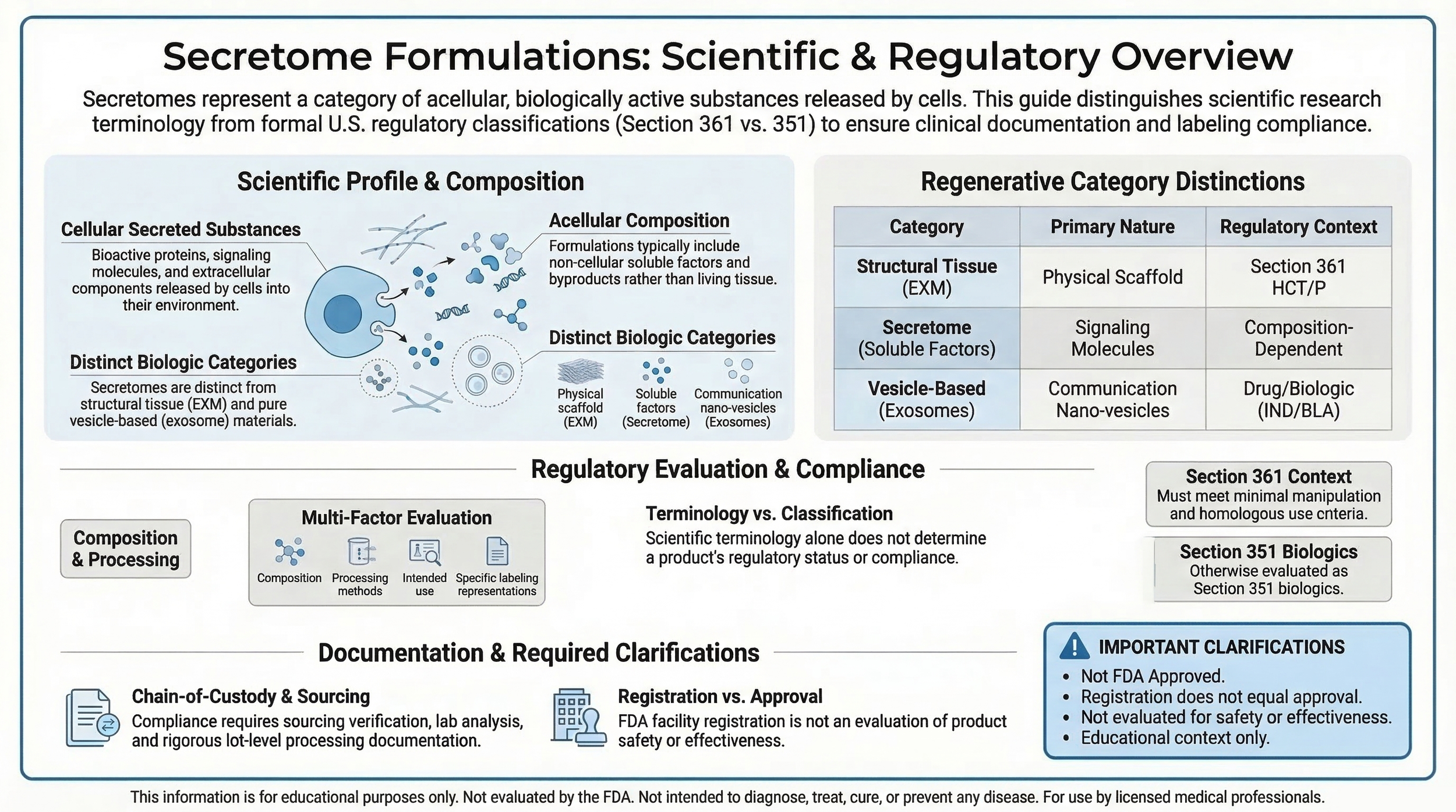
Educational infographic outlining secretome-related formulation characteristics and regulatory classification considerations under federal biologics frameworks.
Secretome formulations refer to acellular preparations described in relation to cellular secretions and extracellular biological components.
This page provides regulatory and classification context for licensed healthcare professionals evaluating sourcing transparency, composition characteristics, and federal positioning considerations under 21 CFR Part 1271 Section 361.
This overview is part of the broader Biologic Product Portfolio .
This material does not describe clinical use, therapeutic outcomes, dosing protocols, or treatment applications.
Scientific Background
The term “secretome” refers broadly to substances released by cells into their surrounding environment. In laboratory research contexts, this may include proteins, signaling molecules, and other extracellular components.
Research discussion of secretome biology does not establish regulatory classification or approved medical use.
Additional scientific context is available within the Peer-Reviewed Scientific References .
Composition & Regulatory Evaluation
Certain formulations described as secretome-based are characterized as non-cellular preparations.
Regulatory evaluation may consider whether a product contains:
- Living cells
- Structural tissue
- Metabolically active cellular components
- Biologically derived soluble factors
Classification depends on composition, processing methodology, labeling representations, intended use, and federal interpretation.
Classification analysis may involve review under minimal manipulation, homologous use, and documentation standards outlined in 21 CFR Part 1271 Subparts A–D.
Federal Regulatory Considerations
Regulatory positioning of secretome-related products depends on product characteristics and interpretation under 21 CFR Part 1271 Section 361 .
Certain products may instead fall under alternative regulatory pathways including biologics licensing requirements under Section 351 of the Public Health Service Act.
Additional context is available within the Section 361 vs Section 351 Overview .
Related Biologic Categories
Professional Evaluation Pathway
Licensed healthcare professionals seeking documentation transparency, sourcing standards, and regulatory positioning clarification may review the structured Structured Supplier Evaluation Process .
Credential confirmation is required prior to documentation release and manufacturer discussion coordination.
Frequently Asked Regulatory Questions
Are secretome formulations approved by the FDA?
No. Use of secretome terminology does not imply FDA approval, clearance, or evaluation of safety or effectiveness.
Do secretome formulations qualify under Section 361?
Qualification under 21 CFR Part 1271 Section 361 requires strict criteria including minimal manipulation and homologous use standards.
Does this page describe clinical use?
No. This material provides regulatory and scientific background only and does not describe treatment protocols, dosing, or outcomes.
Educational reference only. Certain materials discussed may be subject to federal regulation. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease. Federal law restricts distribution to licensed healthcare professionals or by their order.

