Provider Credential Verification Portal
Access to manufacturer documentation, sourcing records, and lot-level traceability materials is restricted to verified licensed healthcare professionals operating within the regulatory framework of 21 CFR Part 1271.
This process operates within the broader framework outlined in the 21 CFR Part 1271 Regulatory Framework and supported by the Regulatory Resource Center.
This portal is not intended for patients, consumers, investors, media inquiries, or general public information requests.
Submission is limited to actively licensed healthcare professionals or authorized institutional representatives. Submissions that do not meet credential verification standards will not be processed.
All submissions are subject to manual review prior to any documentation access coordination.
Regulatory Context
Documentation access operates within the classification structure governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps), including analysis of:
- Section 361 vs Section 351 distinction
- Minimal manipulation interpretation
- Homologous use limitations
- Donor eligibility requirements
- Current Good Tissue Practice (cGTP)
Professional Access & Governance Framework
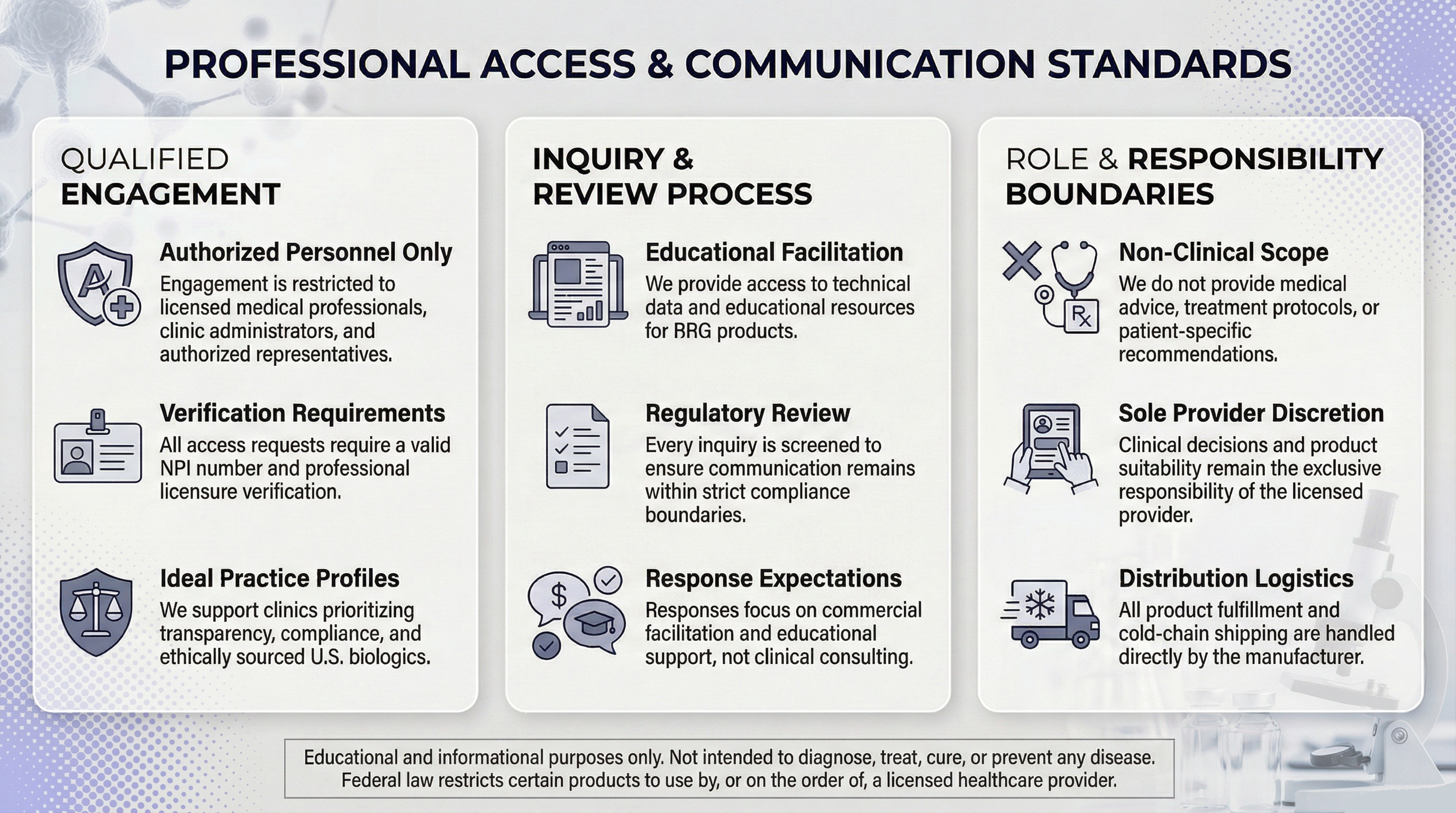
Credential Verification Request
Credential verification is required prior to documentation coordination.
- Credential information is reviewed to verify professional eligibility.
- Review is conducted manually; submission does not result in automatic approval.
- Qualified submissions may receive educational documentation or sourcing transparency materials.
- Requests may be routed to appropriate documentation coordinators depending on the information requested.
- Submission does not guarantee documentation release or supplier relationship.
Next Step — Professional Consultation
For providers seeking structured guidance on regulatory interpretation, documentation review, or sourcing evaluation, a consultation may be requested.
Schedule a Professional Consultation
Organizational Scope
Not By Chance Life LLC operates as a regulatory education and documentation coordination entity and does not manufacture, process, store, or distribute human cells or tissue products.
Submission of a credential verification request does not establish a supplier agreement, purchasing relationship, distribution arrangement, or clinical affiliation. Not By Chance Life LLC provides regulatory education and documentation coordination only and does not provide medical advice, diagnosis, treatment guidance, dosing instruction, or clinical protocols. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness. This information is provided for educational purposes only and is intended for licensed healthcare professionals.

