Current Good Tissue Practice (cGTP) — 21 CFR Part 1271 Subpart D
Current Good Tissue Practice (cGTP) requirements establish manufacturing safeguards applicable to Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
These requirements are codified in 21 CFR Part 1271 Subpart D and operate within the broader regulatory framework defined under 21 CFR Part 1271.
cGTP standards are intended to reduce the risk of communicable disease transmission by requiring appropriate controls across recovery, processing, storage, labeling, packaging, and distribution.
For a structured overview of regulatory criteria and safeguards, see the Regulatory Resource Library.
Documentation and Regulatory Education
Licensed medical professionals and organizations may request educational materials related to regulatory safeguards, quality systems, and documentation practices referenced within this resource.
Credential verification may be required prior to documentation access.
This information is for educational purposes only. Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease. For use by licensed medical professionals.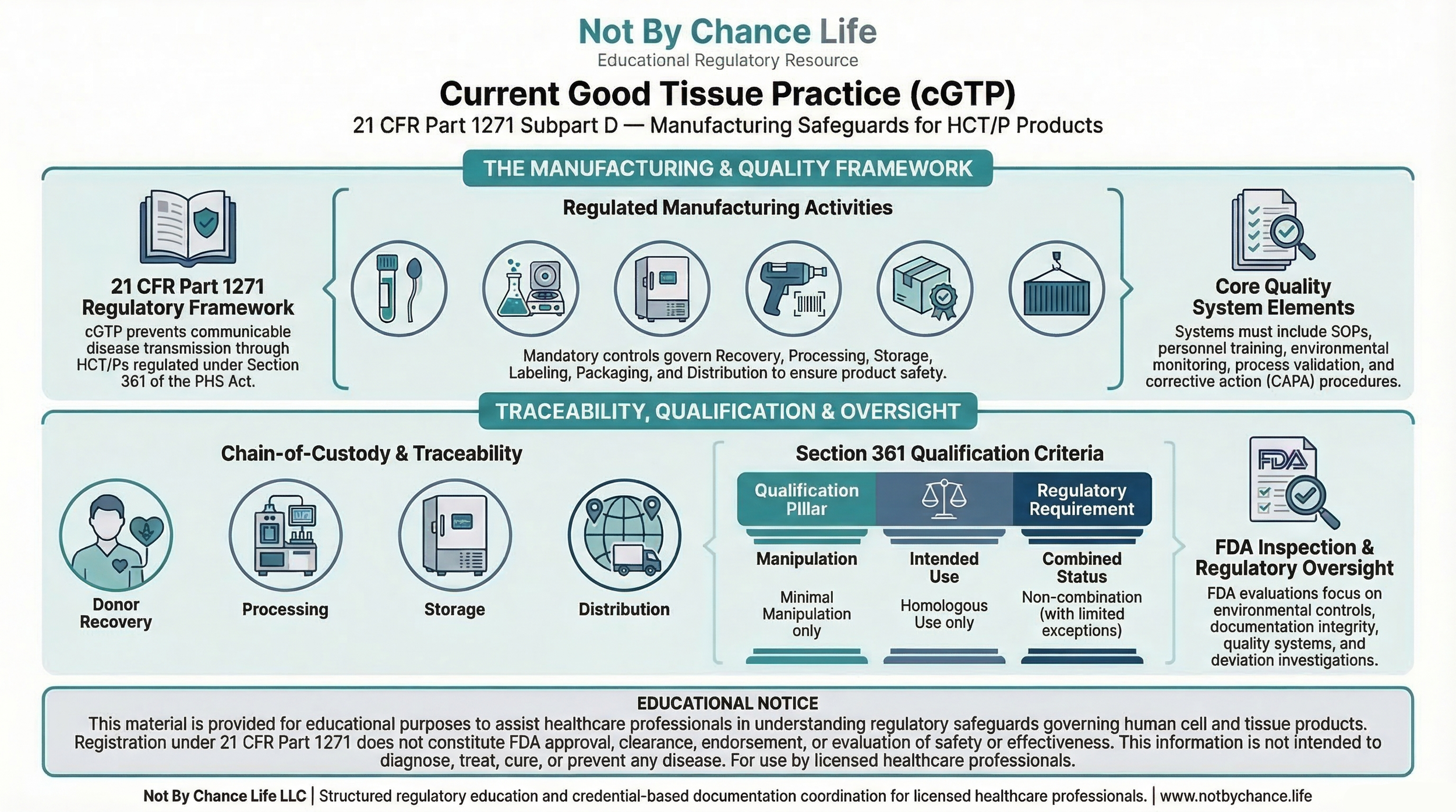
Purpose of cGTP Requirements
- Recovery controls
- Processing controls
- Storage conditions
- Labeling and packaging
- Distribution safeguards
These controls are intended to prevent contamination or cross-contamination of HCT/Ps.
Manufacturing Controls (Subpart D)
- Environmental monitoring
- Equipment maintenance
- Supply controls
- Recordkeeping and traceability
- Complaint handling
Quality System Elements
- Standard operating procedures (SOPs)
- Personnel training
- Traceability systems
- Deviation handling
- Corrective action processes
Role in Section 361 Eligibility
- Minimal Manipulation
- Homologous Use
- Combination status
- Systemic effect considerations
- Donor Eligibility
- cGTP safeguards
Regulatory Oversight
- FDA inspections
- Documentation review
- Traceability verification
- Quality system evaluation
Observations related to cGTP may result in regulatory actions such as warning letters or required corrective measures.
Related Regulatory Topics
Regulatory Notice
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.
Related Regulatory Resources
For additional regulatory clarification and structured interpretation, review the following educational materials:

