Homologous Use Under 21 CFR Part 1271 — Regulatory Standard
Homologous use is a regulatory criterion used by the U.S. Food and Drug Administration (FDA) when evaluating Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
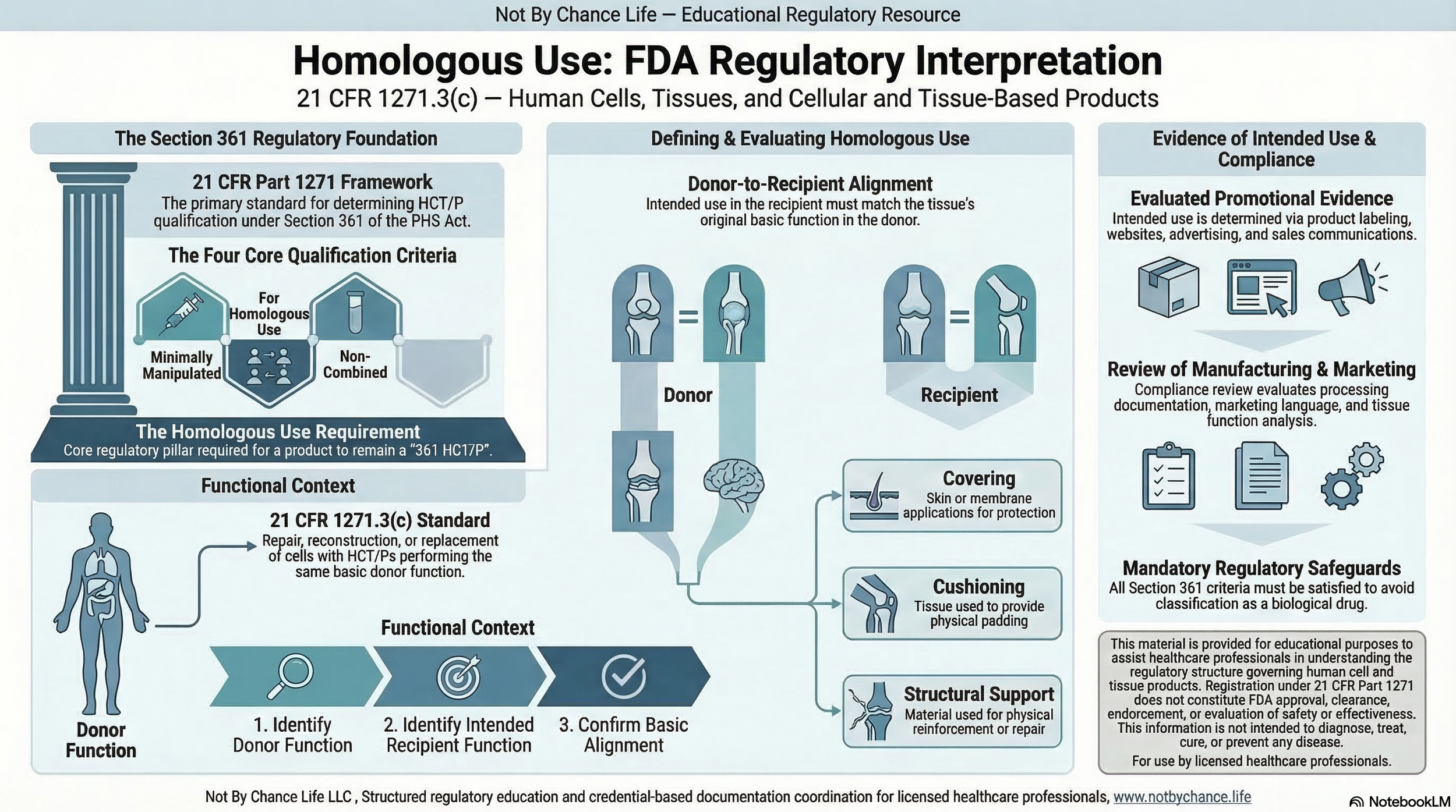
This concept is defined in 21 CFR 1271.3(c) and must be evaluated within the broader framework established under 21 CFR Part 1271.
Homologous use is one of several criteria used to determine whether a product may qualify for regulation solely under Section 361 of the Public Health Service Act.
Regulatory classification criteria are further defined under 21 CFR 1271.10(a) .
It is evaluated alongside minimal manipulation, combination limitations, and systemic effect considerations.
For a structured overview of all regulatory criteria, see the Regulatory Resource Library.
Documentation and Regulatory Education
Licensed medical professionals and organizations may request educational materials related to regulatory criteria, donor eligibility standards, and documentation practices referenced within this resource.
Credential verification may be required prior to documentation access.
Regulatory Definition
Homologous use refers to the repair, reconstruction, replacement, or supplementation of a recipient’s cells or tissues with an HCT/P that performs the same basic function in the recipient as in the donor.
- What is the tissue’s basic structural or biological function?
- What function is intended in the recipient?
- Do those functions align?
For applied interpretation examples: Homologous use interpretation examples →
Intended Use Evaluation
Regulatory evaluation considers objective evidence of intended use, including:
- Labeling
- Website content
- Promotional materials
Function Alignment Considerations
Structural tissues typically provide:
- Covering or barrier function
- Cushioning or support
- Physical structure
Use must remain consistent with these basic functions to meet homologous use criteria.
Role in Regulatory Classification
- Minimal Manipulation
- Combination limitations
- Systemic effect considerations
- Structural vs Cellular Tissue
Related Regulatory Topics
Regulatory Notice
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

