FDA Guidance Documents — HCT/P Regulation
The United States Food and Drug Administration (FDA) publishes guidance documents to assist healthcare professionals, researchers, and regulatory stakeholders in understanding the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
These guidance documents provide regulatory interpretation and compliance clarification related to the regulatory structure established under 21 CFR Part 1271. They help explain how the FDA evaluates regulatory classification, manufacturing practices, and donor eligibility requirements for biological materials derived from human tissue.
The following FDA guidance documents are frequently referenced when evaluating regulatory classification and compliance considerations related to HCT/Ps.
The infographic below summarizes how regulatory agencies and guidance documents help interpret the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products under 21 CFR Part 1271.
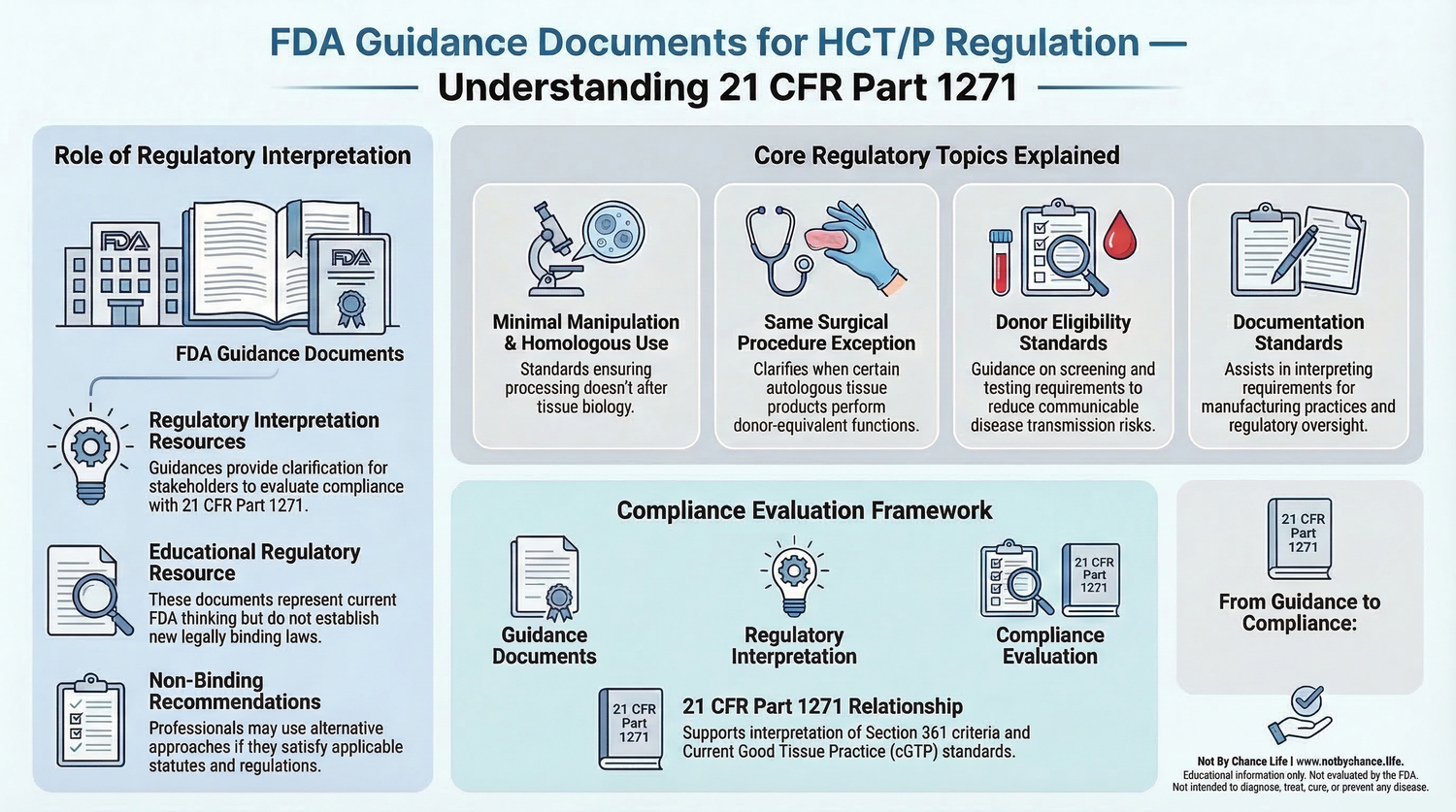
Minimal Manipulation & Homologous Use Guidance
Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use
This FDA guidance document explains how the agency interprets the regulatory concepts of minimal manipulation and homologous use when evaluating whether a product may qualify for regulation solely under Section 361 of the Public Health Service Act.
Official FDA guidance document:
FDA Guidance — Minimal Manipulation & Homologous Use
Educational explanations of these regulatory concepts are available here:
Same Surgical Procedure Exception
Same Surgical Procedure Exception Under 21 CFR 1271.15(b)
This FDA guidance document discusses regulatory interpretation of the same surgical procedure exception. Under certain circumstances, this exception may apply when human cells or tissues are removed from a patient and implanted back into the same patient during the same surgical procedure.
Official FDA guidance document:
HCT/P Regulatory Classification Guidance
Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products
This guidance document provides additional regulatory clarification regarding classification considerations under Section 361 and Section 351 of the Public Health Service Act.
Official FDA guidance document:
FDA Guidance — Regulatory Considerations for HCT/Ps
Educational overview of the regulatory classification framework:
Donor Eligibility Guidance
Eligibility Determination for Donors of Human Cells, Tissues, and Cellular and Tissue-Based Products
FDA guidance documents related to donor eligibility determination describe procedures used to evaluate communicable disease risks associated with tissue donors. These procedures include medical history evaluation, risk assessment, laboratory testing, and documentation of donor eligibility.
Educational explanation of donor eligibility requirements:
This information is provided for educational purposes only.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
Establishment registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
This information is not intended to diagnose, treat, cure, or prevent any disease.

