Donor Eligibility Requirements — 21 CFR 1271 Subpart C
Donor eligibility determination is a regulatory safeguard established under 21 CFR 1271 Subpart C to reduce the risk of communicable disease transmission associated with Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
These requirements operate within the broader regulatory framework defined under 21 CFR Part 1271.
Donor eligibility is one component of the overall regulatory evaluation applied to HCT/Ps, alongside criteria such as minimal manipulation, homologous use, and manufacturing safeguards.
For a structured overview of all regulatory components, see the Regulatory Resource Library.
Documentation and Regulatory Education
Licensed medical professionals and organizations may request educational materials related to donor eligibility requirements, screening standards, and regulatory safeguards referenced within this resource.
Credential verification may be required prior to documentation access.
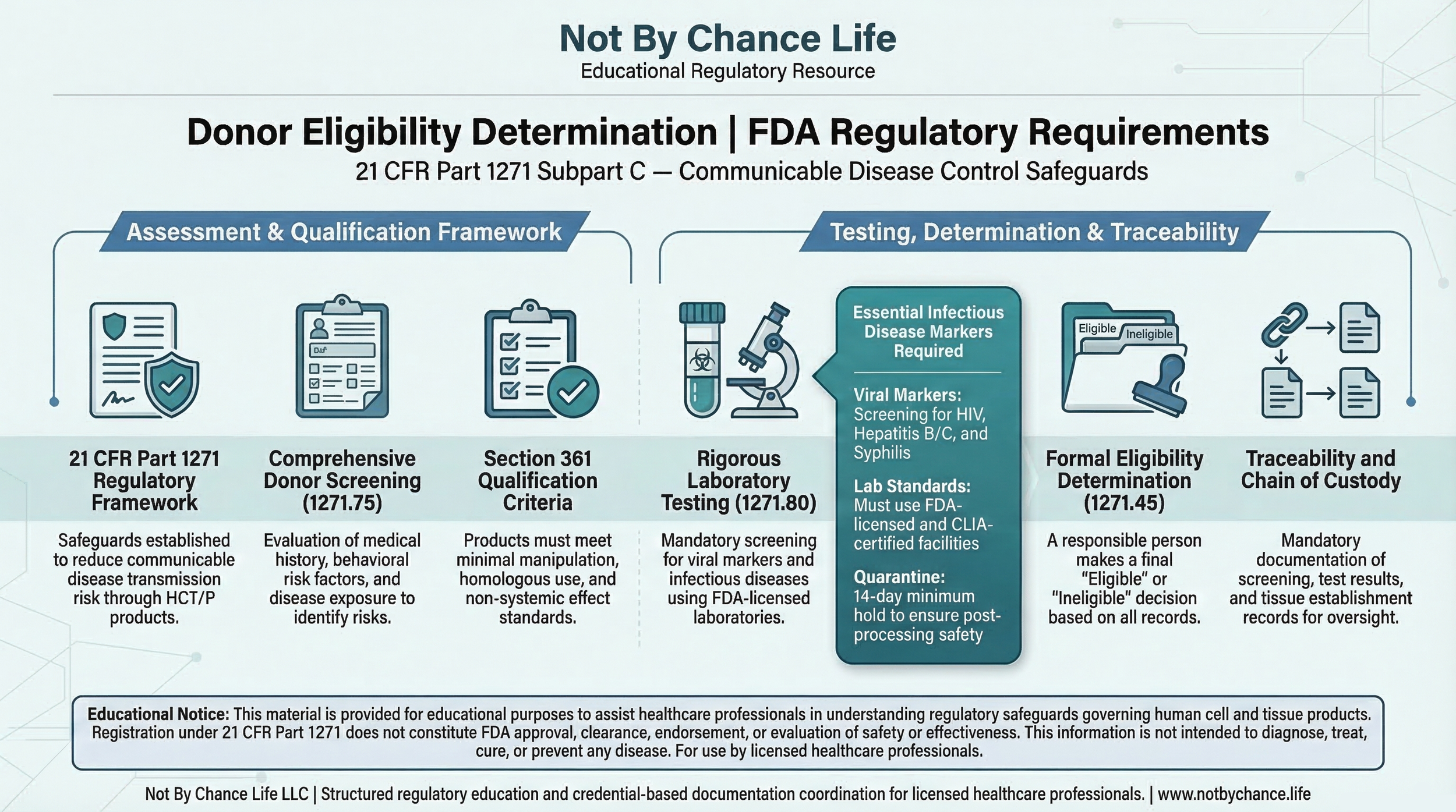
Donor Screening — 21 CFR 1271.75
- Medical history evaluation
- Behavioral risk factor assessment
- Communicable disease exposure review
Donor Testing — 21 CFR 1271.80
Donor testing must be performed using appropriately qualified laboratories and validated testing methodologies for communicable disease markers.
Eligibility Determination — 21 CFR 1271.45
A responsible person must make a formal donor eligibility determination based on screening and testing results in accordance with regulatory criteria.
Role in Regulatory Classification
Additional traceability and documentation safeguards are addressed in Donor Screening, Traceability, and Quality Standards .
Related Regulatory Topics
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

