About Not By Chance Life LLC
Not By Chance Life LLC is a U.S.-based regulatory education and documentation coordination organization supporting licensed medical professionals, multi-location practices, and qualified research institutions evaluating biologic product categories within the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
The organization focuses on improving regulatory literacy and documentation transparency associated with biologic product categories evaluated under federal regulatory criteria including minimal manipulation standards, homologous use interpretation, donor eligibility determination requirements, and current good tissue practice (cGTP) .
Structured documentation review procedures are outlined within the Structured Supplier Evaluation Process .
Category education resources are available within the Biologic Product Portfolio .
Organizational Structure & Role Clarity
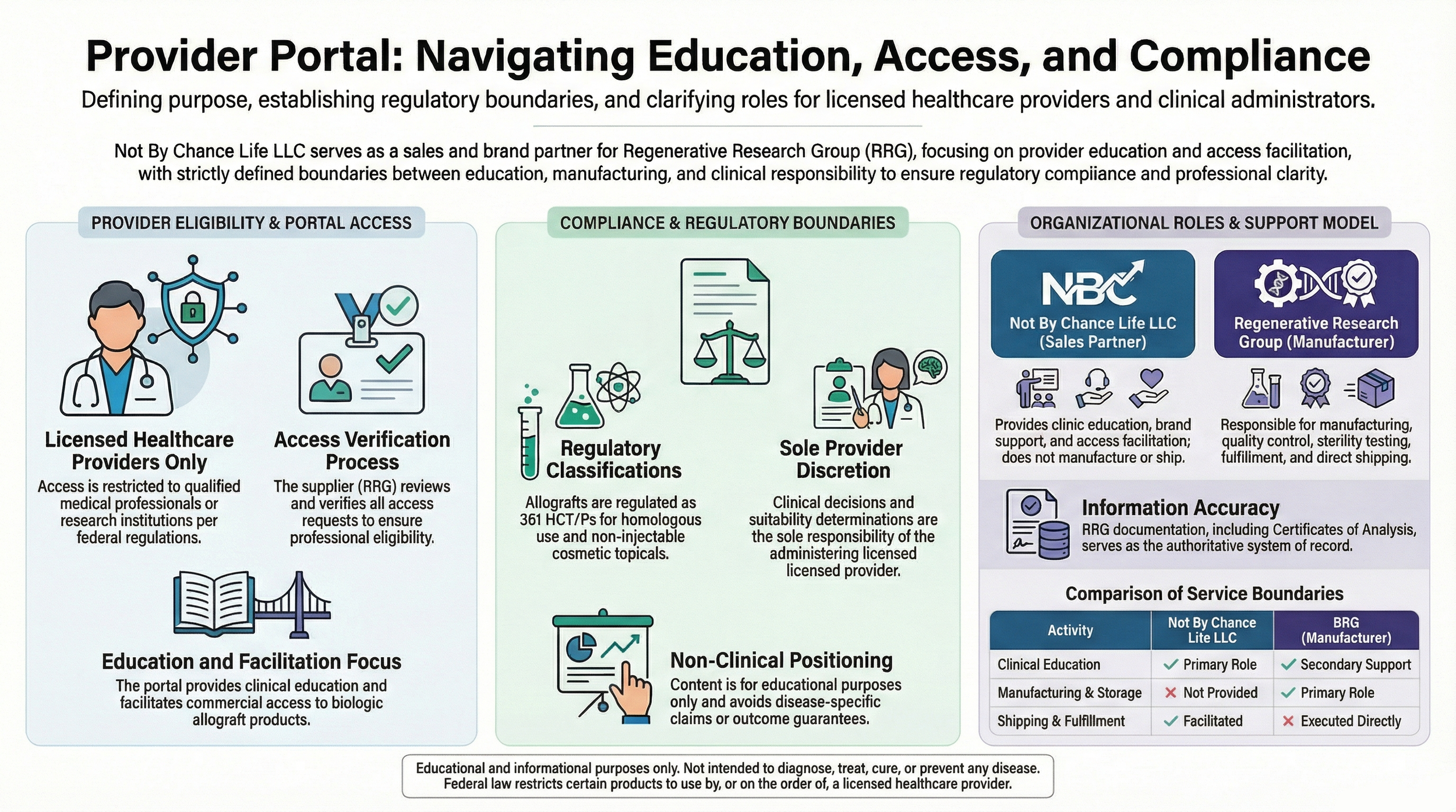
Not By Chance Life LLC operates exclusively as a regulatory education and documentation coordination entity.
The organization does not manufacture, process, store, distribute, or ship biologic materials and does not participate in clinical decision-making or treatment protocol development.
Manufacturing, donor eligibility determination, laboratory testing, chain-of-custody controls, and product fulfillment are performed by licensed tissue establishments operating in accordance with registration and current good tissue practice standards under 21 CFR Part 1271.
Regulatory Alignment & Institutional Discipline
Educational materials are structured to remain consistent with 21 CFR Part 1271 Section 361 , including qualification criteria related to:
- Minimal manipulation regulatory standards
- Homologous use regulatory interpretation
- Combination product limitations
- Systemic effect considerations
- Donor eligibility determination requirements
- Current Good Tissue Practice (cGTP)
Institutional governance safeguards and communication boundaries are outlined in the Governance & Compliance Policy .
Biologic Category Education
Educational overviews are provided for biologic product categories frequently referenced within the HCT/P regulatory framework:
- Extracellular Matrix (EXM) Allograft Overview
- Exosome-Related Formulation Overview
- Secretome Formulation Overview
These educational pages provide sourcing context, composition explanations, and regulatory positioning considerations for professionals conducting independent documentation review and due diligence.
Documentation-First Infrastructure
Responsible evaluation of biologic materials requires structured review of donor screening documentation, sourcing transparency, lot-level traceability, and manufacturer validation standards.
Professionals seeking clarity on documentation review procedures may reference the Structured Supplier Evaluation Process prior to credential submission.
Documentation access requests may be initiated through the Provider Credential Verification Portal .
Professional Audience Limitation
This platform is not consumer-facing and is not intended for patient-directed marketing.
Access to supplier documentation, evaluation materials, and manufacturer resources is restricted to licensed healthcare professionals or by their lawful order in accordance with federal law.
Regulatory Notice
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361 .
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, endorsement, or evaluation of safety or effectiveness.
This information is provided for educational purposes only and is not intended to diagnose, treat, cure, or prevent any disease.

