Minimal Manipulation Under 21 CFR Part 1271 — Regulatory Standard
What is minimal manipulation?
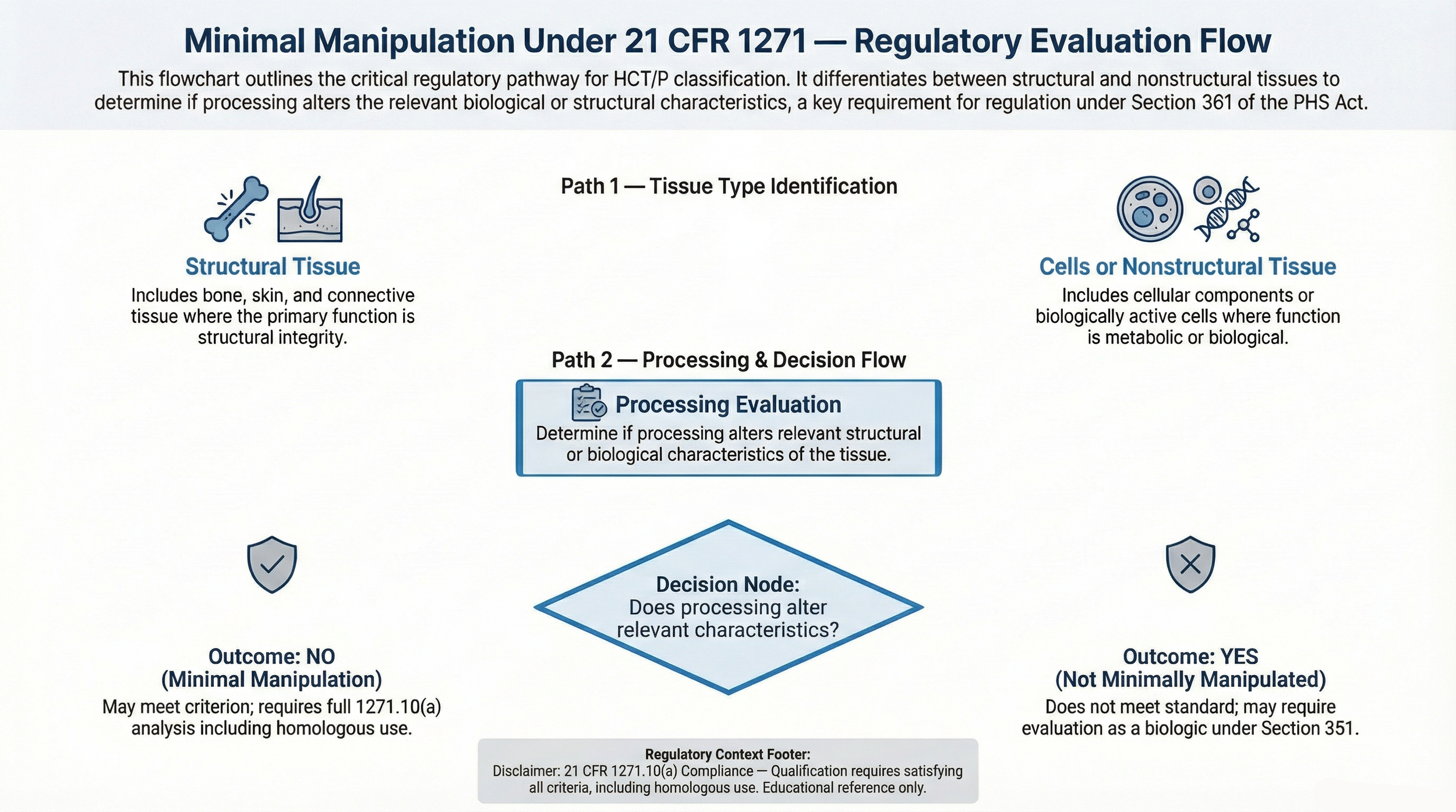
Minimal manipulation refers to processing that does not alter the original relevant characteristics of a human cell or tissue product, as defined under 21 CFR 1271.3(f).
In simple terms, the tissue must retain its original structure or biological characteristics after processing.
Minimal manipulation is a core regulatory criterion used by the U.S. Food and Drug Administration (FDA) when evaluating Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
Licensed medical professionals and organizations seeking regulatory clarity or documentation transparency may request additional educational materials and supporting information through the contact page.
This criterion is defined in 21 CFR 1271.3(f) and must be evaluated within the broader framework established under 21 CFR Part 1271.
Regulatory classification criteria are further defined under 21 CFR 1271.10(a) .
Minimal manipulation is one of several requirements used to determine whether a product may qualify for regulation solely under Section 361 of the Public Health Service Act.
These criteria are evaluated collectively under 21 CFR 1271.10(a) classification criteria .
Inconsistent classification or incomplete documentation may influence how biologic products are evaluated under applicable regulatory standards.
Documentation and Regulatory Education
Not sure how these requirements apply to your current sourcing or protocols?
Licensed medical professionals may request structured documentation review and regulatory overview materials prior to engaging with biologic suppliers.
This process supports documentation clarity, classification alignment, and sourcing transparency under 21 CFR Part 1271.
Credential verification is required prior to documentation access.
Request Regulatory Information
Provider Credential Verification Portal
Schedule a Professional Consultation
Requests are reviewed to confirm professional credentials and determine appropriate documentation access pathways.
Regulatory Definition
- Structural tissue: Processing that does not alter the original relevant characteristics relating to its utility for support, covering, or structural function.
- Cells or nonstructural tissues: Processing that does not alter relevant biological characteristics.
Structural Tissue Considerations
- Support and cushioning
- Barrier or covering function
- Connective structure
Cellular and Nonstructural Tissue Considerations
- Biological activity
- Metabolic characteristics
- Cellular function
Role in Regulatory Classification
- Minimal manipulation
- Homologous use
- Combination limitations
- Systemic effect considerations
For applied interpretation examples: Homologous use interpretation →
Regulatory Notice
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

