Governance & Compliance Policy
This Governance & Compliance Policy defines the regulatory framework under which educational materials and biologic product categories referenced on this platform are discussed.
The purpose of this policy is to establish regulatory alignment, communication discipline standards, documentation safeguards, credential controls, and professional responsibility consistent with applicable federal and state law.
Regulatory analysis referenced throughout this platform may involve Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) governed under 21 CFR Part 1271 and Section 361 of the Public Health Service Act.
Governance & Compliance Framework Overview
The visual summary below outlines regulatory alignment structure, communication safeguards, traceability controls, and clinical autonomy boundaries.
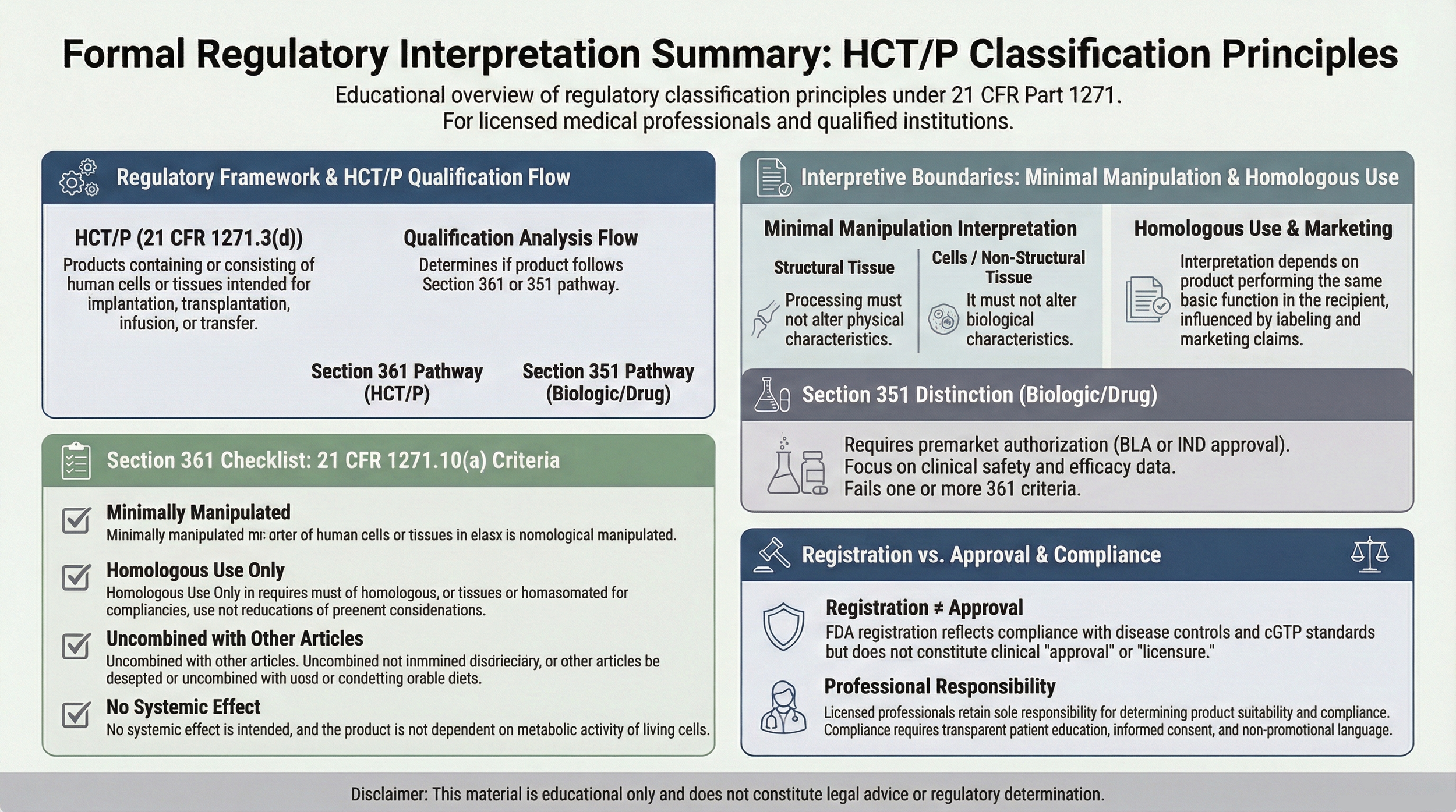
Regulatory Alignment
Certain product categories referenced may qualify for regulation solely under Section 361 if they meet eligibility criteria outlined in 21 CFR 1271.10(a), including minimal manipulation and homologous use standards.
Establishments operating under 21 CFR Part 1271 are subject to registration, donor eligibility determination (Subpart C), and Current Good Tissue Practice (cGTP) manufacturing safeguards (Subpart D).
Establishment registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
Communication Standards
All communications referencing tissue-based products must remain consistent with regulatory classification boundaries.
The following representations are prohibited:
- Statements implying FDA approval, clearance, or endorsement
- Claims to diagnose, treat, cure, or prevent disease
- Guarantees of specific patient outcomes
- Non-homologous use positioning inconsistent with Section 361 eligibility
Marketing language and educational materials are structured to maintain regulatory neutrality and avoid therapeutic claims.
Documentation & Traceability Standards
HCT/Ps regulated under 21 CFR Part 1271 are subject to documented donor eligibility determination, communicable disease screening, and cGTP quality controls.
Documentation frameworks may include:
- Donor eligibility determination records
- Communicable disease laboratory verification
- Lot identification and traceability logs
- Processing and preservation documentation
- Quality system and deviation reporting controls
Licensed professionals are responsible for reviewing available documentation and maintaining appropriate clinical records consistent with regulatory requirements.
Credential Verification & Distribution Controls
Federal law restricts distribution of certain tissue-based products to licensed healthcare professionals or by their order.
Credential verification processes may be implemented prior to granting access to detailed product documentation, lot records, and compliance materials.
Distribution safeguards support alignment with federal regulatory structure and manufacturer governance protocols.
Clinical Autonomy & Independent Judgment
Licensed healthcare professionals retain full responsibility for independent clinical evaluation, patient selection, procedural decisions, and regulatory compliance.
No content on this platform should be interpreted as prescribing, directing, or mandating clinical practice.
Educational materials are not a substitute for professional medical or legal judgment.
Audit & Review Posture
Public-facing communications and educational materials are periodically reviewed for alignment with applicable regulatory frameworks, including 21 CFR Part 1271 and related FDA guidance.
Updates may be implemented to reflect evolving regulatory interpretation, enforcement trends, or compliance clarification.
Shared Compliance Responsibility
Compliance requires coordinated alignment among recovery establishments, processing facilities, distributors, and licensed healthcare professionals.
Providers remain responsible for ensuring that use, communication, and documentation remain consistent with federal and state regulatory requirements.
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Establishment registration does not constitute FDA approval. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

