Regulatory Education & Documentation Transparency for HCT/P Evaluation
Not By Chance Life LLC provides structured regulatory education and documentation review frameworks for licensed healthcare professionals evaluating Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under the 21 CFR Part 1271 regulatory framework.
Educational materials focus on regulatory classification criteria, documentation transparency, donor eligibility considerations, and supplier evaluation context within the federal framework governing human tissue products.
For structured regulatory review or documentation evaluation guidance, professionals may request a consultation or proceed to credential verification.
Documentation Before Supplier Engagement
The following educational reference diagram summarizes key regulatory considerations frequently evaluated when reviewing supplier documentation within the HCT/P regulatory framework, including criteria defined under 21 CFR 1271.10(a).
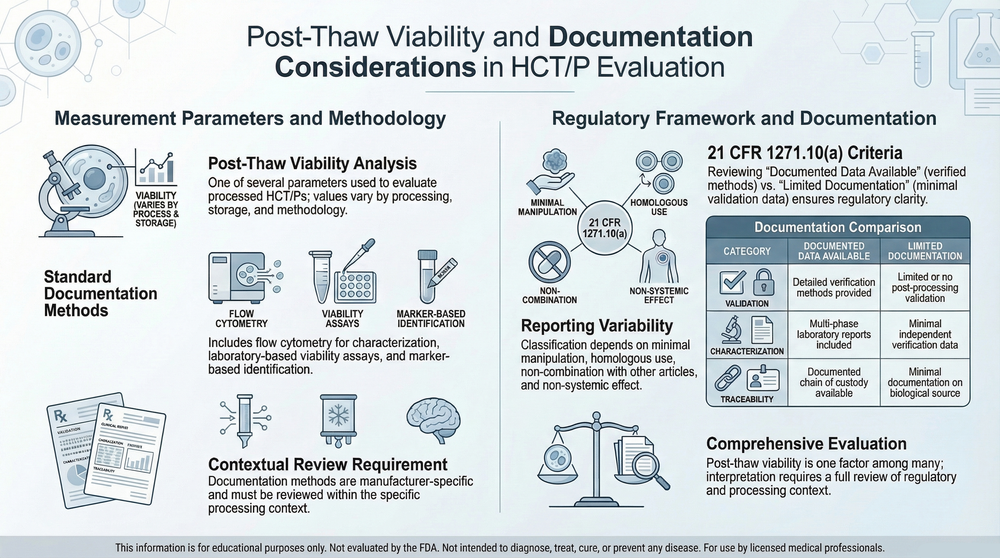
Aligned with evaluation criteria outlined in 21 CFR 1271.10(a).
See how this fits into structured evaluation: Regulatory Evaluation Pathway →
Start Here — Core Regulatory Framework
For foundational understanding of regulatory classification and evaluation criteria, begin with the primary framework and core concepts below:
Professional Consultation
Licensed healthcare professionals seeking structured regulatory clarification, documentation review guidance, or supplier evaluation alignment may request a consultation.
Professional Credential Review
Access to manufacturer documentation, supplier records, and structured evaluation materials is restricted to licensed healthcare professionals and authorized clinical entities.
Regulatory & Compliance Notice
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
This information is provided for educational purposes only and is not intended to diagnose, treat, cure, or prevent any disease.

