Donor Screening, Traceability & Quality Standards
Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) regulated under 21 CFR Part 1271 must follow defined regulatory controls for donor eligibility determination, communicable disease testing, and operational quality standards.
This page provides an educational overview of donor screening, traceability requirements, Certificate of Analysis (COA) documentation, and Current Good Tissue Practice (cGTP) operational standards within the framework defined by 21 CFR 1271 Subpart C.
For a broader regulatory overview, see the Regulatory Resource Library.
Donor Eligibility Determination — 21 CFR 1271 Subpart C
Donor eligibility determination under 21 CFR 1271 Subpart C is a required regulatory safeguard intended to reduce the risk of communicable disease transmission through HCT/Ps and forms part of Section 361 compliance criteria.
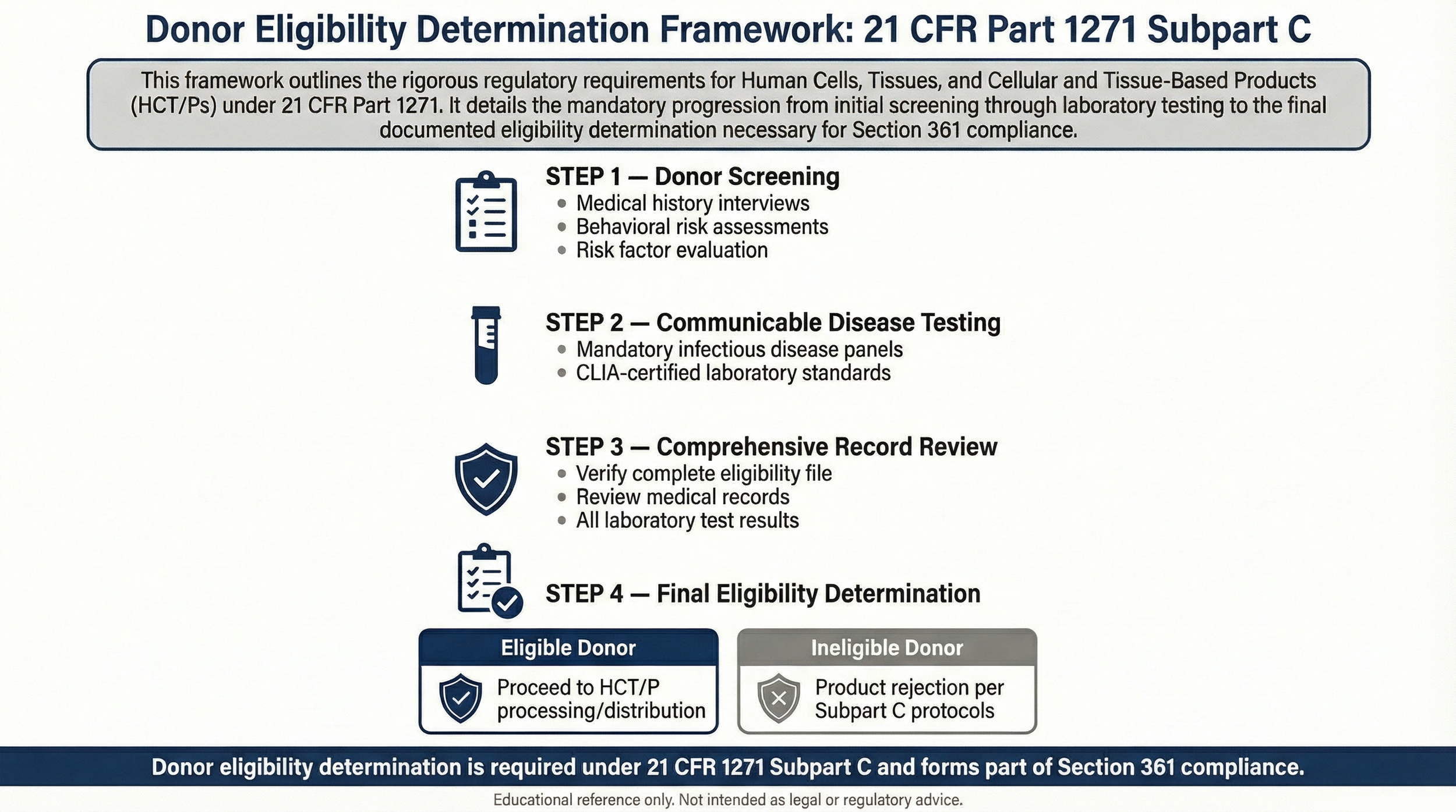
Aligned with regulatory safeguards supporting Section 361 HCT/P classification under 21 CFR 1271.
- Medical history review
- Behavioral risk assessment
- Communicable disease risk evaluation
- Required laboratory testing
A detailed regulatory explanation is available in our Donor Eligibility Requirements — 21 CFR 1271 Subpart C reference page.
Communicable Disease Testing Requirements
Communicable disease testing requirements under 21 CFR 1271.80 require testing to be performed by appropriately certified laboratories using validated methodologies.
These testing standards support the communicable disease control pathway that distinguishes Section 361 HCT/Ps from products regulated under Section 351.
Additional regulatory context is outlined in Section 361 vs Section 351 classification .
Current Good Tissue Practice (cGTP)
Current Good Tissue Practice (cGTP) requirements under 21 CFR 1271 Subpart D establish operational controls for HCT/P establishments involved in recovery, processing, storage, and distribution.
- Facility environmental control
- Equipment validation
- Personnel training
- Deviation investigation procedures
Operational safeguards are explained in our Current Good Tissue Practice overview .
Chain-of-Custody & Traceability
Traceability requirements ensure that tissue recovery, processing, storage, and distribution can be tracked through documented lot identification systems and chain-of-custody records.
- Lot identification numbers
- Recovery site documentation
- Processing records
- Distribution tracking
Certificate of Analysis (COA)
A Certificate of Analysis (COA) may document sterility testing, identity verification, and additional quality parameters associated with tissue processing and lot release.
A COA does not represent FDA approval but may support documentation review within a cGTP-compliant framework.
This information is for educational purposes only. Certain materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness. Not intended to diagnose, treat, cure, or prevent any disease. For use by licensed healthcare professionals.

