HCT/P Regulatory Evaluation Pathway
Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) are regulated in the United States under the framework established in 21 CFR Part 1271. Regulatory classification depends on whether a product meets the eligibility criteria described in 21 CFR 1271.10(a).
Understanding the regulatory evaluation pathway helps explain how products may be regulated either under Section 361 of the Public Health Service Act or under the biologics licensing framework associated with Section 351.
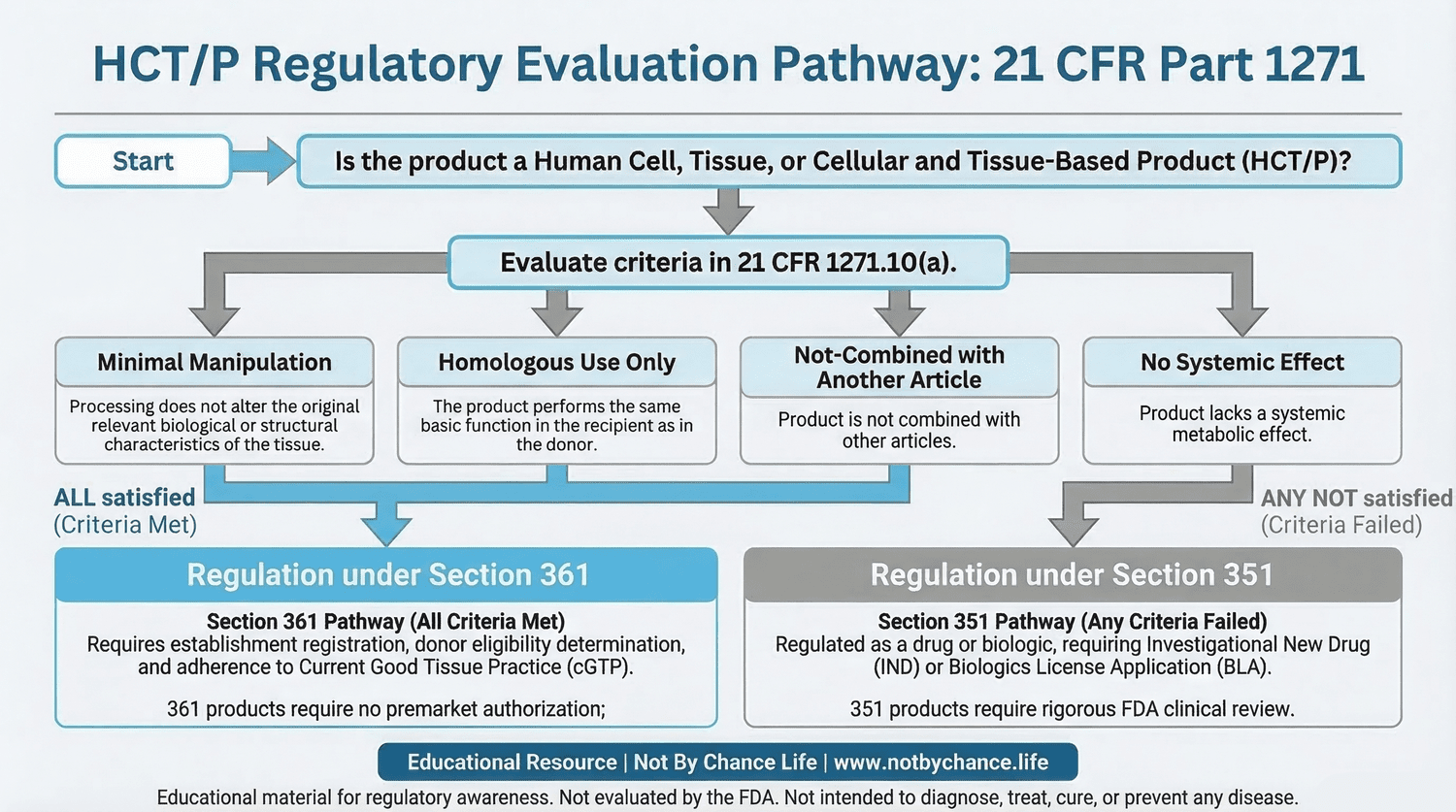
Educational diagram illustrating the regulatory evaluation pathway used when assessing how Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) may be classified under 21 CFR Part 1271.
Initial Regulatory Evaluation
When evaluating an HCT/P product, several regulatory criteria may be examined to determine whether the product may qualify for regulation solely under Section 361.
- Minimal Manipulation
- Homologous Use
- whether the product is combined with another article
- whether systemic effects are intended
If all regulatory criteria described in 21 CFR 1271.10(a) are satisfied, the product may fall within the Section 361 regulatory framework.
Section 361 Regulatory Pathway
Products that meet all criteria in 21 CFR 1271.10(a) may be regulated solely under Section 361 of the Public Health Service Act.
Establishments handling these products remain subject to regulatory safeguards that may include donor eligibility determination, establishment registration, and compliance with Current Good Tissue Practice (cGTP) requirements.
Section 351 Regulatory Pathway
If an HCT/P product does not meet the criteria outlined in 21 CFR 1271.10(a), the product may be regulated as a biological product under Section 351 of the Public Health Service Act.
Products regulated under Section 351 may require additional regulatory oversight, which may include biologics license applications and other regulatory review processes.
Regulatory Criteria Frequently Examined
- Minimal Manipulation — 21 CFR 1271.3(f)
- Homologous Use — 21 CFR 1271.3(c)
- Structural Tissue vs Cellular Tissue
- Donor Eligibility Determination
- Current Good Tissue Practice (cGTP)
- FDA Enforcement & Compliance
Importance of Understanding the Evaluation Framework
Healthcare professionals, regulatory specialists, and tissue establishments may review the regulatory evaluation framework when assessing compliance considerations related to human cell and tissue products.
Educational resources describing these regulatory concepts can assist organizations in understanding the regulatory environment surrounding HCT/P products and the safeguards intended to reduce communicable disease transmission.
This information is provided for educational purposes only.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
This information is not intended to diagnose, treat, cure, or prevent any disease.

