FDA Inspection Process for HCT/P Establishments
The United States Food and Drug Administration (FDA) conducts inspections of certain establishments involved in activities related to Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
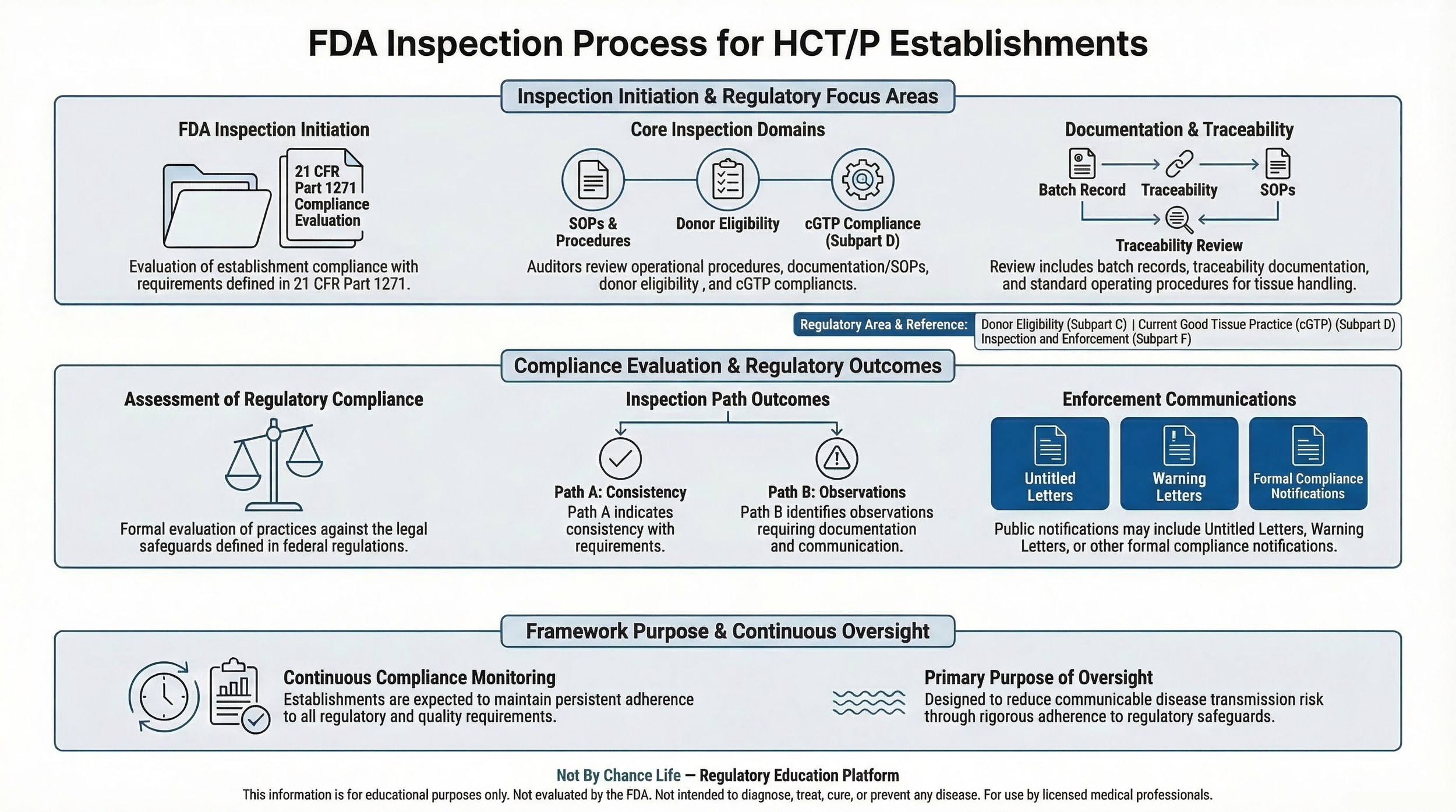
These inspections help evaluate compliance with regulatory requirements established under 21 CFR Part 1271.
Inspection activities may include review of operational procedures, documentation practices, and compliance with regulatory safeguards intended to reduce the risk of communicable disease transmission associated with human tissue products.
Purpose of FDA Inspections
FDA inspections are intended to assess whether establishments handling human tissue products are operating in accordance with applicable regulatory requirements.
Inspection activities may include review of procedures related to:
- tissue recovery and procurement
- processing procedures
- storage and distribution practices
- recordkeeping and documentation
- compliance with Current Good Tissue Practice (cGTP)
Current Good Tissue Practice (cGTP)
Operational safeguards governing HCT/P handling are defined in 21 CFR Part 1271 Subpart D.
These requirements establish standards for tissue processing, storage, labeling, packaging, and distribution.
Additional educational explanation is available in Current Good Tissue Practice (cGTP).
Inspection Findings and Regulatory Communication
Inspection findings may lead to regulatory communications if concerns are identified regarding compliance with applicable regulations.
These communications may include observations related to operational procedures, documentation practices, or regulatory interpretation.
Public enforcement communications related to HCT/P regulation are discussed in FDA Warning Letters — HCT/P Regulatory Enforcement.
Professional Documentation Access
Licensed healthcare professionals and qualified institutions evaluating HCT/P regulatory compliance may request structured documentation related to inspection readiness, supplier qualification, and regulatory review processes.
Documentation review may include:
- Inspection readiness and compliance framework summaries
- cGTP quality system documentation (Subpart D)
- Donor eligibility verification processes (Subpart C)
- Traceability and chain-of-custody documentation
- Standard operating procedures and documentation controls
- Regulatory interpretation and compliance review support
Request Professional Information
Access is limited to licensed medical professionals and qualified organizations.
Request Documentation ReviewThis information is for educational purposes only.
Establishment registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

