Understanding 21 CFR Part 1271 — Key Concepts
This page provides an educational overview of key concepts within 21 CFR Part 1271, the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
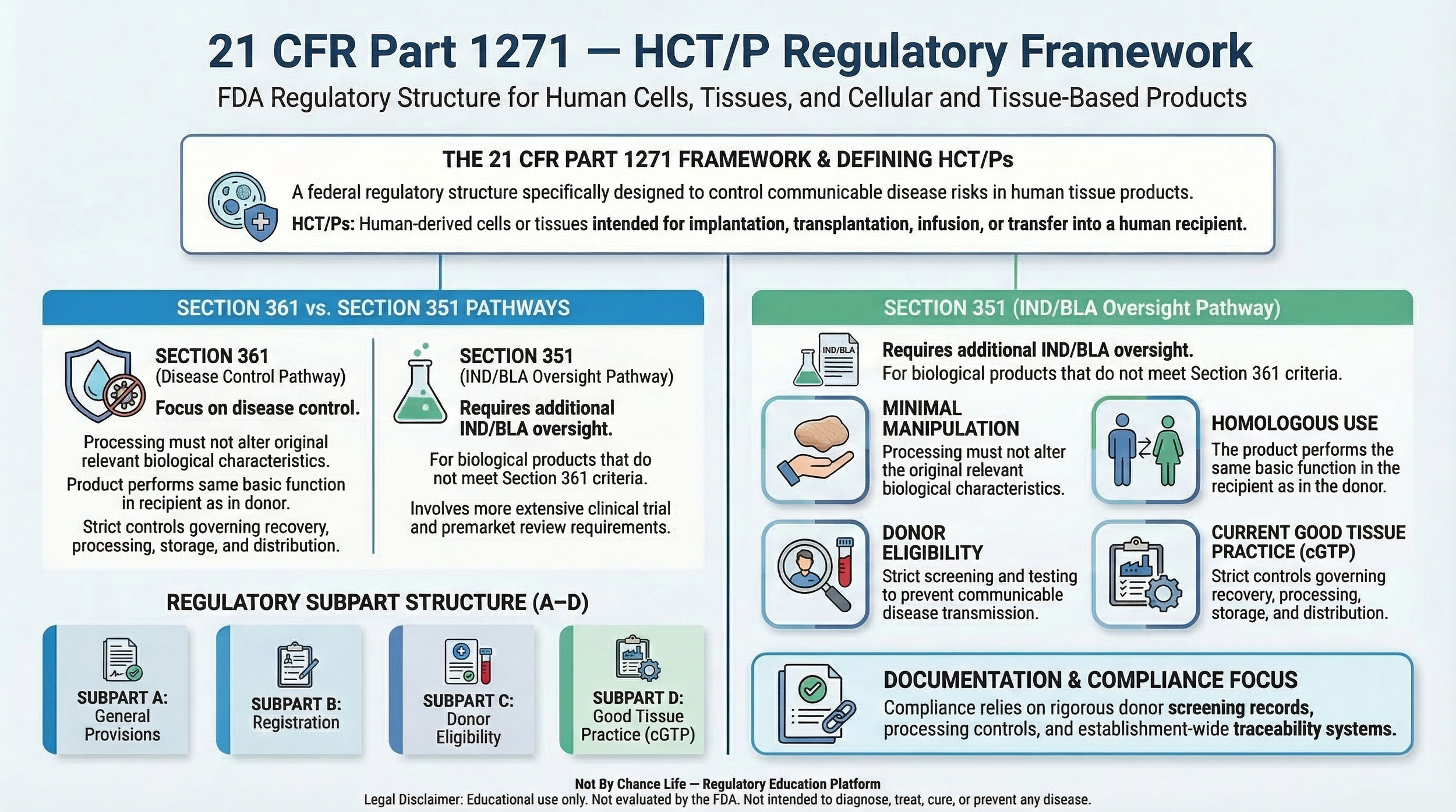
Key regulatory criteria under 21 CFR Part 1271 include Section 1271.10(a) classification requirements, homologous use, minimal manipulation, and donor eligibility standards, which are evaluated collectively to determine appropriate regulatory classification.
For the complete regulatory framework and full classification criteria, refer to: 21 CFR Part 1271 Regulatory Framework.
What This Regulation Covers
21 CFR Part 1271 establishes requirements for the evaluation, processing, storage, labeling, and distribution of human tissue products, with a primary focus on communicable disease risk control and regulatory classification.
Core Regulatory Concepts
Regulatory classification under 21 CFR Part 1271 is based on defined criteria. Each concept below contributes to determining whether a product may qualify under Section 361.
- Section 361 vs Section 351 Classification
- Minimal Manipulation
- Homologous Use
- Donor Eligibility
- Current Good Tissue Practice (cGTP)
- Structural vs Cellular Tissue
- Regulatory Subparts A–D
- Regulatory Glossary
How Products Are Evaluated
HCT/Ps are evaluated under criteria defined in 21 CFR 1271.10(a) to determine whether they may be regulated solely under Section 361 of the Public Health Service Act.
Detailed classification considerations are outlined in: 21 CFR 1271.10(a) classification criteria →
- Minimal manipulation
- Homologous use
- Combination with other articles
- Systemic effect considerations
A structured review of these criteria is required when evaluating classification and regulatory positioning.
For a step-by-step evaluation framework: View Regulatory Evaluation Pathway
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.

