FDA Warning Letters — HCT/P Regulatory Enforcement
The United States Food and Drug Administration (FDA) publishes warning letters and other enforcement communications when the agency identifies regulatory concerns related to human biological products.
Public enforcement communications can provide insight into how regulatory authorities interpret and apply federal regulations governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
This page provides an educational overview of regulatory enforcement patterns frequently referenced in publicly available warning letters related to HCT/P regulatory compliance.
Key Regulatory Themes in FDA Warning Letters
FDA enforcement communications involving HCT/P regulation frequently reference several regulatory criteria used to evaluate product classification and regulatory compliance.
- Minimal manipulation requirements
- Homologous use interpretation
- Product classification under Section 361 vs Section 351
- Manufacturing and processing controls
- Current Good Tissue Practice (cGTP) requirements
Understanding these regulatory criteria can help professionals interpret enforcement communications and evaluate compliance considerations within the HCT/P regulatory framework.
The infographic below summarizes how FDA warning letters and regulatory enforcement communications are commonly evaluated within the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products under 21 CFR Part 1271.
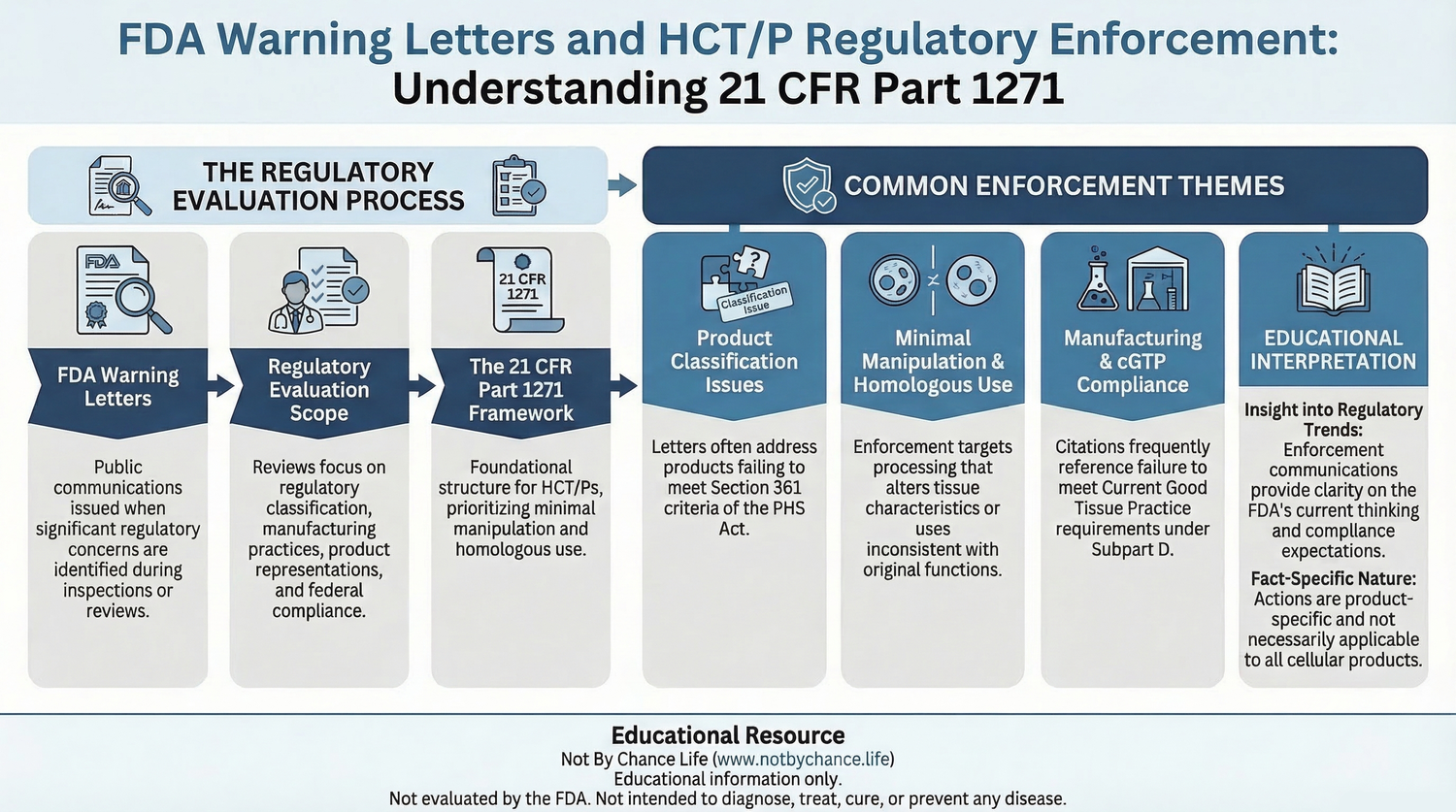
Regulatory Issues Frequently Referenced in Warning Letters
Public enforcement communications involving HCT/P regulation often reference several regulatory criteria used to evaluate product classification and regulatory compliance.
- Minimal manipulation requirements
- Homologous use interpretation
- Product classification under Section 361 vs Section 351
- Manufacturing and processing controls
- Current Good Tissue Practice (cGTP) requirements
Educational discussions of these regulatory concepts are available in the following pages:
- Minimal Manipulation Under 21 CFR 1271
- Homologous Use Interpretation
- Section 361 vs Section 351 Regulatory Comparison
- Current Good Tissue Practice Overview
Examples of Enforcement Themes
Product Classification Concerns
Some warning letters discuss situations in which the FDA determined that a biological product did not satisfy the regulatory criteria required to qualify solely under Section 361 of the Public Health Service Act.
In such cases the agency may determine that the product is subject to regulation as a biological product under the Public Health Service Act and related drug or biologics regulatory pathways.
Minimal Manipulation Interpretation
Enforcement communications may reference situations where processing methods were determined to alter the original relevant characteristics of structural tissue or the biological characteristics of cells or non-structural tissue.
These determinations can influence regulatory classification under 21 CFR Part 1271.
Homologous Use Interpretation
Some enforcement communications reference intended use representations that the agency determined did not satisfy the homologous use criterion defined in 21 CFR 1271.3(c).
Manufacturing and Processing Controls
Regulatory communications may also reference operational concerns involving manufacturing safeguards, documentation practices, or compliance with current good tissue practice requirements under 21 CFR Part 1271 Subpart D.
Educational Value of Enforcement Communications
Public enforcement communications can provide insight into regulatory interpretation trends and compliance expectations within the HCT/P regulatory framework.
However, individual enforcement actions are fact-specific and should not be interpreted as general regulatory conclusions applicable to all biological products or regulatory situations.
Understanding the context of enforcement communications may assist regulatory professionals, healthcare institutions, and compliance reviewers when evaluating biologic materials and documentation practices.
Educational reference only. Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361.
Establishment registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
This information is not intended to diagnose, treat, cure, or prevent any disease.

