FDA Tissue Establishment Registration (21 CFR Part 1271 Subpart B)
Organizations involved in the recovery, processing, storage, labeling, packaging, or distribution of certain human cell and tissue products may be required to register with the U.S. Food and Drug Administration (FDA) as a tissue establishment.
Registration requirements are outlined in 21 CFR Part 1271 Subpart B, which governs establishment registration and product listing for Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
The purpose of establishment registration is to enable FDA oversight and tracking of organizations participating in the handling and distribution of regulated tissue products.
21 CFR Part 1271 Registration Framework Summary
The FDA requires certain organizations handling human cells or tissues to register as tissue establishments under 21 CFR Part 1271 Subpart B. Registration allows the agency to maintain visibility into facilities involved in the recovery, processing, storage, labeling, packaging, or distribution of Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
- Establishment Registration — organizations must submit facility information to the FDA.
- Product Listing — establishments identify HCT/P products handled by the organization.
- Manufacturing Activity Disclosure — includes processing, testing, storage, labeling, and distribution activities.
- Regulatory Oversight — registration supports FDA inspection and compliance monitoring programs.
Establishment registration is a regulatory reporting requirement and does not represent FDA product approval, clearance, endorsement, or evaluation of safety or effectiveness.
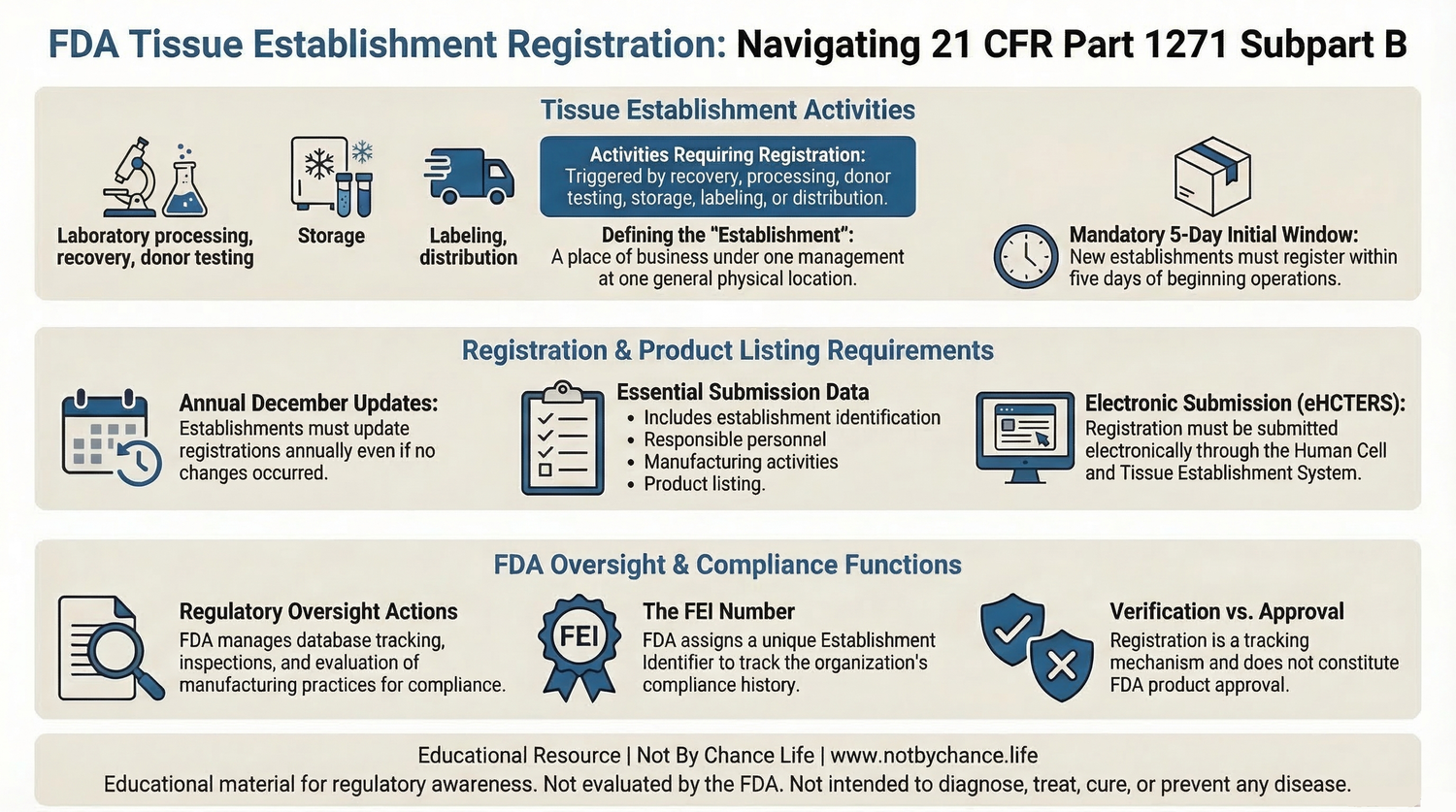
Educational infographic summarizing FDA tissue establishment registration requirements under 21 CFR Part 1271 Subpart B. Educational resource provided by Not By Chance Life.
What Is a Tissue Establishment?
Under the regulatory framework defined in 21 CFR 1271.3(e), a tissue establishment generally refers to any organization that performs one or more manufacturing steps involving human cells or tissues.
These activities may include:
- tissue recovery
- processing or preparation
- testing and screening
- storage
- labeling and packaging
- distribution
Entities performing these functions may fall within FDA registration requirements depending on the nature of the activities performed.
Purpose of Establishment Registration
FDA tissue establishment registration allows the agency to maintain visibility into organizations participating in the HCT/P supply chain.
Registration enables regulatory oversight of:
- tissue processing activities
- donor screening and testing practices
- compliance with current good tissue practice (cGTP)
- distribution of regulated tissue products
Registration and Product Listing
Organizations subject to registration requirements must submit information to FDA identifying the establishment and listing the HCT/Ps handled by the organization.
Registration submissions typically include:
- establishment identification information
- contact information for responsible personnel
- description of activities performed
- listing of HCT/P products handled by the establishment
Registrations are generally submitted through FDA’s electronic registration system.
Relationship to Other Regulatory Requirements
Establishment registration alone does not determine the regulatory classification of a product. Regulatory status depends on whether the product meets the criteria outlined in 21 CFR 1271.10(a).
Key criteria include:
- minimal manipulation
- homologous use
- non-combination with certain articles
- limited systemic effect
Further explanation of these criteria is available in the following pages:
- Minimal Manipulation
- Homologous Use
- Section 361 vs Section 351 Classification
- 21 CFR 1271 Regulatory Framework
- FDA HCT/P Guidance Documents
This information is provided for educational purposes only. Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, endorsement, or evaluation of safety or effectiveness. This information is not intended to diagnose, treat, cure, or prevent any disease.

