FDA HCT/P Establishment Registration Requirements
Organizations involved in the recovery, processing, storage, labeling, packaging, or distribution of human cell and tissue products may be subject to establishment registration requirements under 21 CFR Part 1271. These regulations govern Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
Establishment registration is part of the FDA regulatory framework designed to maintain oversight of facilities involved in the handling of biologic materials derived from human tissue.
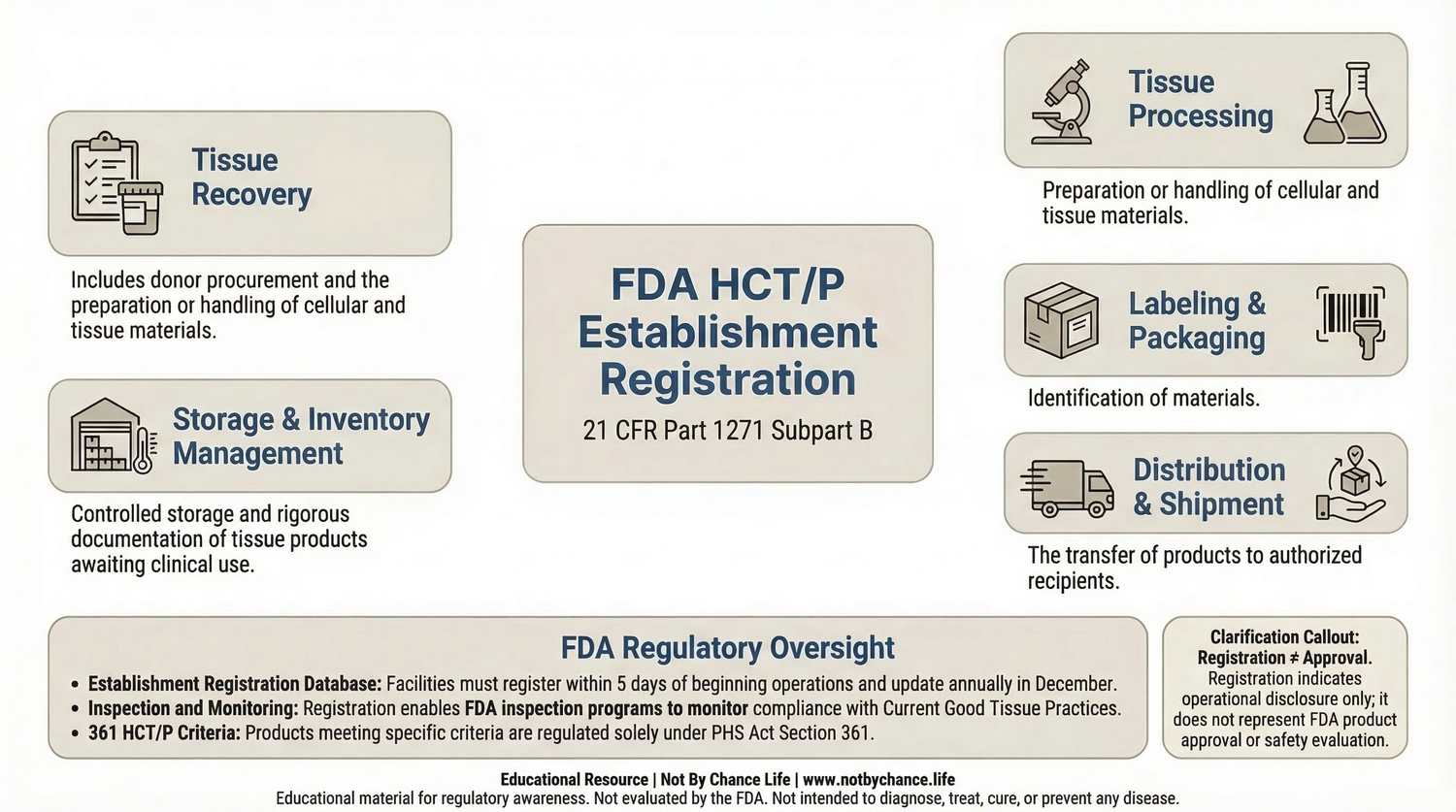
Educational diagram illustrating the regulatory framework for FDA HCT/P establishment registration under 21 CFR Part 1271 Subpart B and the activities that may require registration.
Purpose of Establishment Registration
FDA establishment registration allows the agency to maintain a database of tissue establishments engaged in activities involving HCT/P products. This system supports regulatory oversight, inspection programs, and monitoring of manufacturing practices.
Registration helps the agency identify facilities involved in the handling of human tissues and evaluate compliance with applicable regulatory requirements.
Entities That May Be Required to Register
Under 21 CFR Part 1271 Subpart B, certain organizations involved in HCT/P activities may be required to register with the FDA. These activities may include:
- recovery of human tissues
- processing of tissue products
- storage of tissue materials
- labeling or packaging of HCT/P products
- distribution of human tissue products
Specific regulatory obligations depend on the nature of the activities performed and whether the products meet the criteria described in 21 CFR 1271.10(a).
Relationship Between Registration and Regulatory Approval
Establishment registration with the FDA does not represent product approval, clearance, endorsement, or evaluation of safety or effectiveness.
Registration indicates that a facility has submitted required information to the FDA regarding its operations involving HCT/P products.
Additional Compliance Requirements
In addition to registration requirements, tissue establishments may also be subject to regulatory provisions including:
- Donor Eligibility Determination
- Current Good Tissue Practice (cGTP)
- recordkeeping and traceability documentation
- inspection programs and regulatory oversight
Further explanation of these regulatory components is available in the following pages:
Role of Establishment Registration in Regulatory Oversight
The establishment registration framework supports the FDA's ability to monitor facilities involved in the handling and distribution of human tissues. It also provides transparency regarding organizations operating within the HCT/P regulatory environment.
Healthcare professionals and organizations evaluating biologic materials may review establishment registration status when assessing regulatory documentation.
Related Regulatory Education Resources
- 21 CFR Part 1271 Regulatory Framework
- Minimal Manipulation
- Homologous Use
- Structural Tissue vs Cellular Tissue
- Tissue Chain of Custody
- Regulatory Resource Library
This information is provided for educational purposes only.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, endorsement, or evaluation of safety or effectiveness.
This information is not intended to diagnose, treat, cure, or prevent any disease.

