Tissue Chain of Custody Documentation
Traceability and documentation are essential components of regulatory oversight for Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps). The regulatory framework described in 21 CFR Part 1271 includes requirements designed to ensure that human tissue products can be traced from donor recovery through processing, storage, distribution, and final use.
Chain-of-custody documentation supports transparency and accountability within the tissue handling process and is an important component of Current Good Tissue Practice (cGTP).
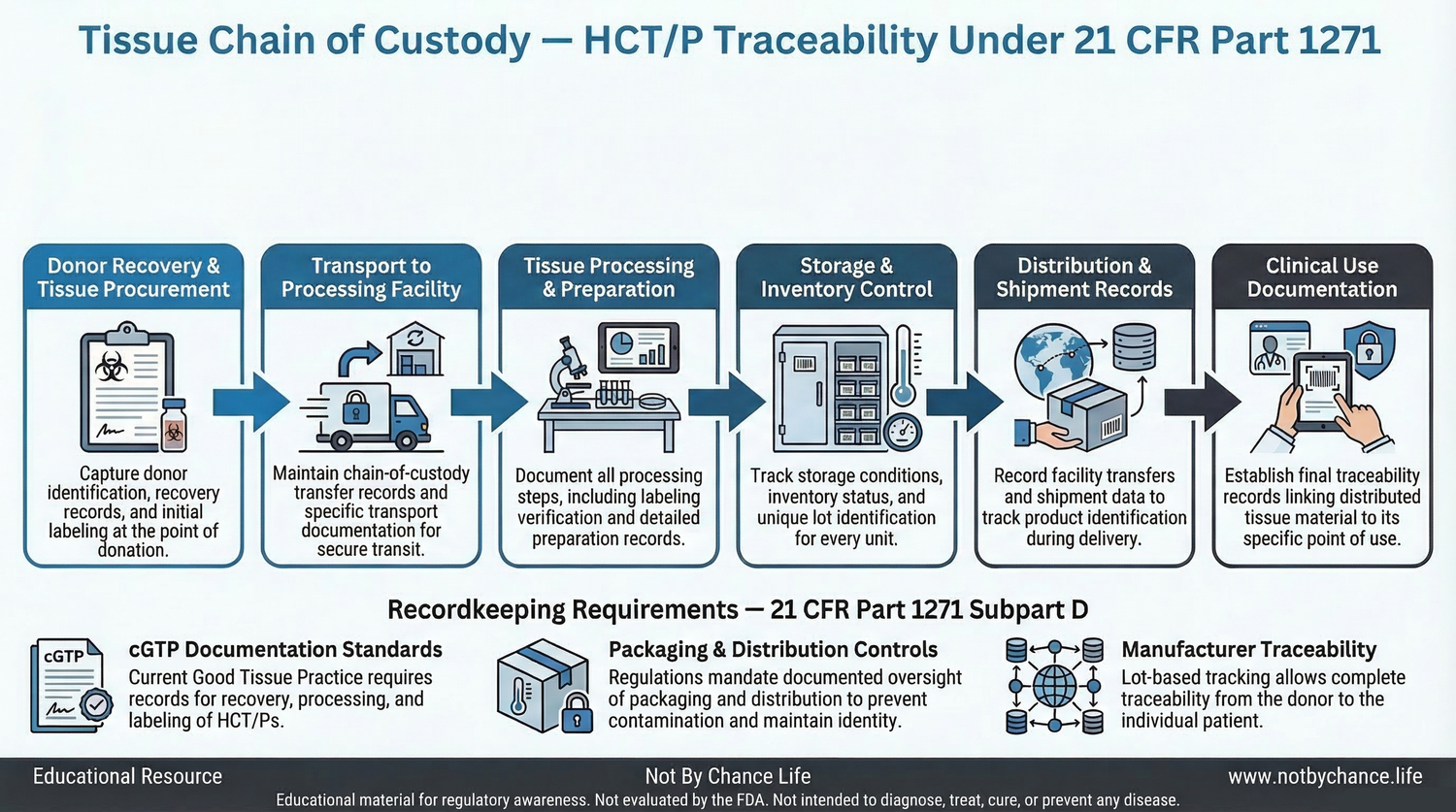
Educational diagram illustrating the chain-of-custody traceability pathway for Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under the regulatory framework described in 21 CFR Part 1271.
Purpose of Chain-of-Custody Documentation
Chain-of-custody documentation establishes a traceable record of how human tissue materials move through each stage of handling and processing. These records help maintain accountability and assist regulatory authorities in monitoring compliance with applicable regulations.
Traceability systems help ensure that tissue materials are properly identified and documented throughout their lifecycle.
Stages in the Tissue Traceability Process
A typical chain-of-custody record for tissue products may include documentation related to several stages of handling:
- donor recovery and tissue procurement
- transport to processing facilities
- tissue processing and preparation
- storage and inventory control
- distribution and shipment records
- documentation associated with clinical use
Maintaining records across these stages helps ensure that tissue materials remain identifiable and traceable throughout the supply chain.
Relationship to Current Good Tissue Practice (cGTP)
Current Good Tissue Practice regulations outlined in 21 CFR Part 1271 Subpart D include recordkeeping requirements designed to support traceability of human cell and tissue products.
These requirements help ensure that tissue establishments maintain documentation related to recovery, processing, storage, labeling, packaging, and distribution of HCT/P products.
Importance of Traceability in Regulatory Oversight
Traceability systems support regulatory oversight by allowing organizations and regulatory authorities to identify how tissue products were handled and distributed. These records can assist in monitoring compliance and maintaining transparency within the tissue supply chain.
Healthcare organizations and regulatory professionals may review chain-of-custody documentation when evaluating biologic materials subject to federal oversight.
Related Regulatory Education Pages
- 21 CFR Part 1271 Regulatory Framework
- Minimal Manipulation — 21 CFR 1271.3(f)
- Homologous Use — 21 CFR 1271.3(c)
- Donor Eligibility Requirements
- Current Good Tissue Practice (cGTP)
- HCT/P Establishment Registration
- Regulatory Resource Library
This information is provided for educational purposes only.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, endorsement, or evaluation of safety or effectiveness.
This information is not intended to diagnose, treat, cure, or prevent any disease.

