Placental Tissue Allograft Overview
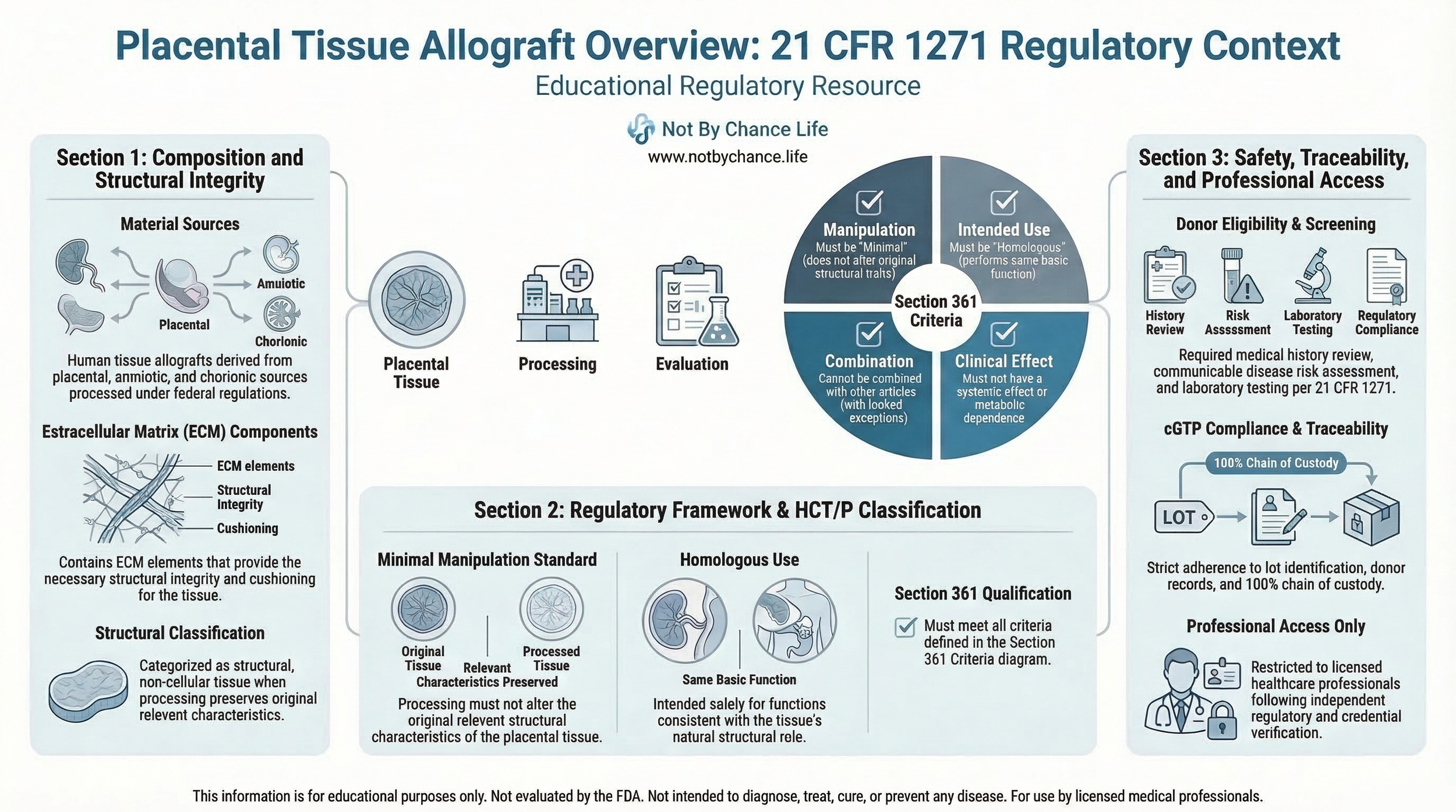
Educational infographic outlining placental tissue allograft characteristics and regulatory evaluation considerations under 21 CFR Part 1271.
Placental tissue allografts refer to human tissue materials derived from placental sources and processed in accordance with federal tissue regulations. These materials may include structural components such as amniotic or chorionic tissue when handled under compliant recovery and processing standards.
This page provides an educational overview of placental tissue allograft characteristics, sourcing requirements, documentation standards, and regulatory considerations relevant to licensed medical professionals and qualified institutions.
Structural Composition
Placental tissues contain structural extracellular matrix components that contribute to their physical integrity. When processed to remove cellular components, certain placental-derived materials may be categorized as structural, non-cellular allografts.
Regulatory review of these materials typically evaluates whether processing preserves the tissue’s original relevant structural characteristics.
Donor Eligibility & Screening
Placental tissue recovery must comply with donor eligibility determination requirements under 21 CFR Part 1271. These requirements include medical history review, communicable disease risk assessment, and laboratory testing.
Compliance with donor screening standards is intended to reduce the risk of communicable disease transmission and support traceability protocols.
Processing & Minimal Manipulation
Processing methods are evaluated under the concept of minimal manipulation, as outlined in 21 CFR 1271 Section 361.
For structural tissues, minimal manipulation generally means that processing does not alter the original relevant structural characteristics of the tissue.
Classification outcomes depend on specific processing methods, labeling context, and intended use considerations.
HCT/P Classification Considerations
Placental tissue allografts may qualify for regulation solely under Section 361 if they satisfy all applicable criteria, including:
- Minimal manipulation
- Homologous use
- No combination with other articles (except permitted exceptions)
- No systemic effect or metabolic dependence, unless qualifying for specific allowances
Additional explanation of classification pathways is available in the HCT/P Classification Overview.
Traceability & Documentation
Establishments involved in recovery, processing, storage, labeling, packaging, or distribution must comply with current good tissue practice (cGTP) requirements.
Documentation typically includes lot identification, donor eligibility records, and chain-of-custody procedures to maintain traceability.
Relationship to Extracellular Matrix (EXM)
Certain placental tissue materials contain extracellular matrix components that may be referenced in structural tissue contexts. Additional educational information is available in the Extracellular Matrix (EXM) Overview .
Educational Context
This information is for educational purposes only.
Not evaluated by the FDA. Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed medical professionals.
Reference Materials
For regulatory reference, consult:
Educational reference only. Certain products may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR 1271 Section 361. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease. Federal law restricts distribution to licensed medical professionals or by their order.

