Regulatory Review & Source Methodology
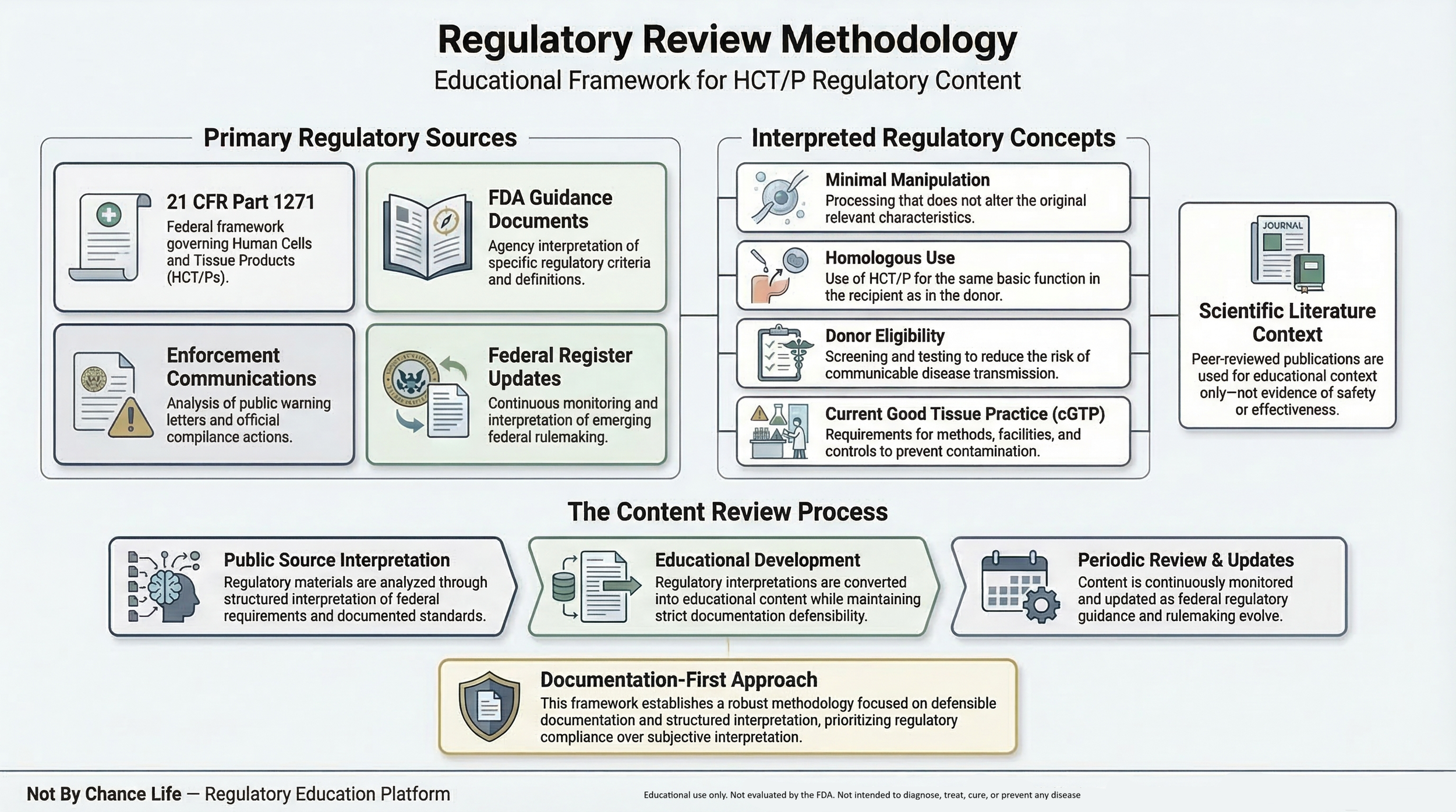
Educational materials published by Not By Chance Life are developed using publicly available regulatory sources related to the United States regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
The purpose of this page is to explain the research sources and review framework used when developing educational regulatory content. For foundational context, see the HCT/P Regulatory Framework Overview.
Primary Regulatory Sources
Educational materials related to HCT/P regulation are primarily based on publicly available regulatory documents including:
- 21 CFR Part 1271 — Human Cells, Tissues, and Cellular and Tissue-Based Products
- FDA guidance documents addressing regulatory interpretation of HCT/P criteria
- FDA public enforcement actions including warning letters and compliance communications
- Federal Register publications describing regulatory updates
These regulatory materials establish the federal framework governing donor eligibility determination, communicable disease safeguards, and current good tissue practice (cGTP) requirements.
Regulatory Interpretation Framework
Educational discussions on this website focus on explaining regulatory terminology commonly referenced in federal tissue regulations, including:
- Minimal manipulation
- Homologous use
- Structural tissue classification
- Donor eligibility determination
- Current Good Tissue Practice (cGTP)
- Traceability and documentation safeguards
These concepts are defined within 21 CFR Part 1271 and related FDA regulatory guidance documents. A structured breakdown is available in 21 CFR Part 1271 Subparts A–D.
Scientific Literature Context
Certain educational materials may reference peer-reviewed scientific publications related to tissue biology, extracellular matrix research, and related areas of biological science.
These references are provided for educational context only and should not be interpreted as evidence of safety, effectiveness, or clinical benefit for any specific product, biologic material, or medical procedure. Additional context is available within peer-reviewed scientific references.
Content Review & Updates
Educational materials are periodically reviewed and updated as regulatory guidance documents, statutory frameworks, or relevant regulatory interpretations evolve.
Updates may incorporate additional publicly available regulatory materials or peer-reviewed literature where appropriate. For structured evaluation standards, see the Structured Supplier Evaluation Process.
This information is for educational purposes only.
Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
Not intended to diagnose, treat, cure, or prevent any disease.
For use by licensed healthcare professionals.

