Transparency & Ethical Sourcing Statement
This statement outlines the principles governing sourcing, documentation, regulatory alignment, and professional responsibility in connection with biologic products discussed on this platform.
The intent is to provide licensed medical professionals and qualified institutions with clarity regarding ethical standards, traceability controls, and compliance positioning.
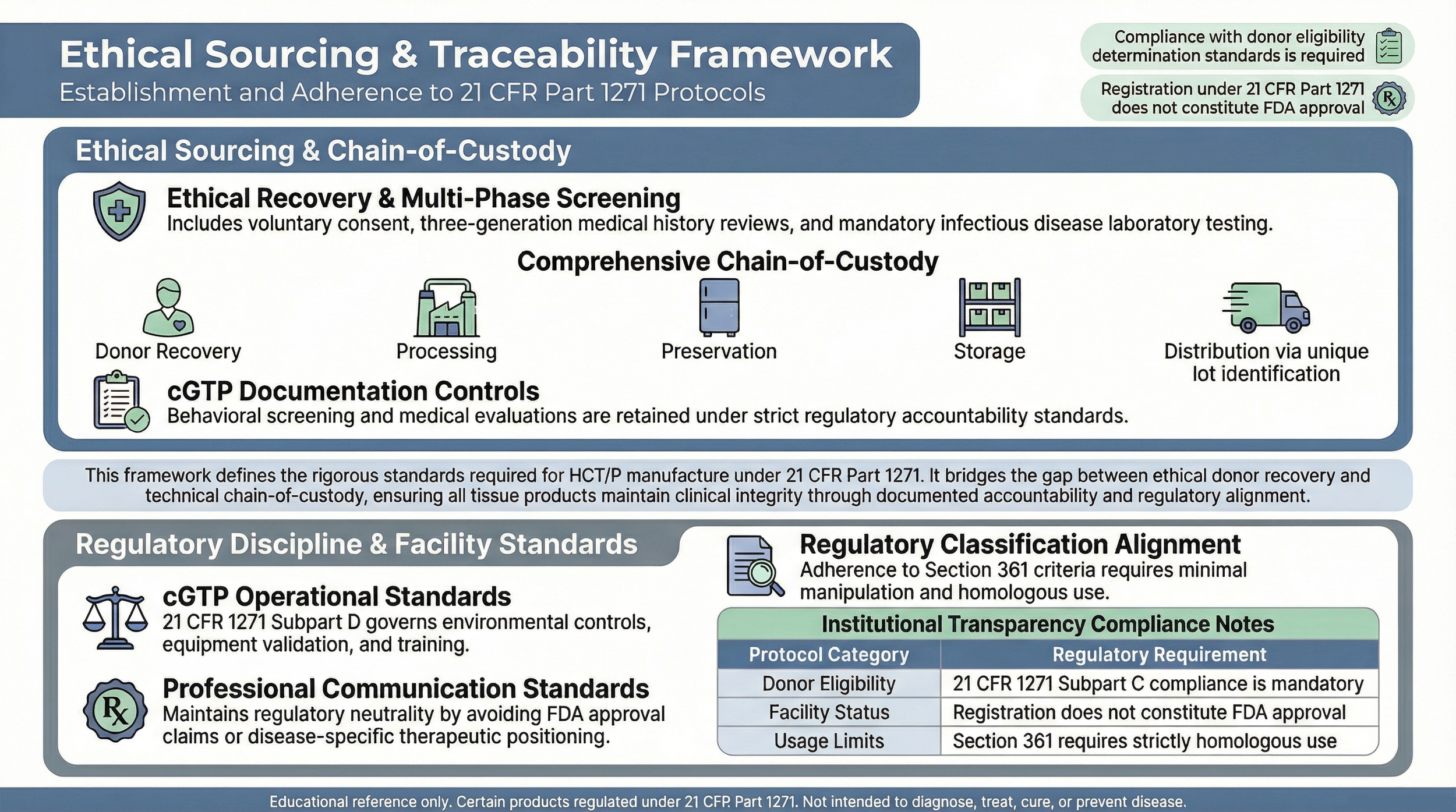
Ethical Tissue Recovery Standards
Tissue materials referenced in educational materials are expected to originate from voluntary donor programs operating under applicable federal and state requirements.
Donor eligibility determinations must comply with 21 CFR 1271 Subpart C , including medical history review, communicable disease risk assessment, and required laboratory testing.
Recovery procedures should be performed in accordance with established medical standards and documented chain-of-custody protocols.
Informed Consent & Donor Screening
Ethical sourcing requires documented informed consent prior to tissue recovery. Donors should undergo comprehensive screening, including medical, behavioral, and laboratory evaluation, consistent with regulatory requirements.
Documentation of donor eligibility and screening must be maintained in accordance with current good tissue practice (cGTP) standards .
Traceability & Chain-of-Custody
Traceability systems are essential for maintaining accountability from recovery through processing, storage, and distribution.
These systems may include:
- Unique lot identification numbers
- Recovery site documentation
- Processing and preservation records
- Storage and transport logs
Proper documentation supports quality oversight and regulatory accountability.
Processing & Facility Controls
Establishments handling HCT/Ps must comply with current good tissue practice (cGTP) requirements under 21 CFR 1271 Subpart D .
These standards address environmental controls, equipment validation, personnel training, deviation reporting, and complaint management.
Registration under 21 CFR Part 1271 does not constitute FDA approval or endorsement but requires adherence to these operational standards.
Regulatory Alignment
Products referenced may qualify for regulation solely under Section 361 if all applicable criteria are satisfied, including minimal manipulation and homologous use.
Products that do not meet these criteria may require alternative regulatory pathways, including regulation under Section 351 of the Public Health Service Act .
Licensed professionals remain responsible for independent regulatory evaluation and compliance with applicable laws.
Commitment to Responsible Communication
Communications regarding tissue-based products should avoid representations that imply FDA approval for diagnosing, treating, curing, or preventing specific diseases.
Educational materials are structured to align with regulatory classification and homologous use limitations.
Professional Responsibility
Ethical sourcing and regulatory compliance require coordination among recovery establishments, processing facilities, distributors, and licensed medical professionals.
Providers are responsible for independent due diligence, documentation retention, and communication consistent with federal and state requirements.
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR 1271. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

