Regulatory Position Statement
This Regulatory Position Statement outlines the framework under which biologic products referenced on this platform are discussed and positioned for licensed medical professionals and qualified institutions.
The intent of this statement is to clarify regulatory boundaries, classification considerations, and professional responsibilities consistent with federal law.
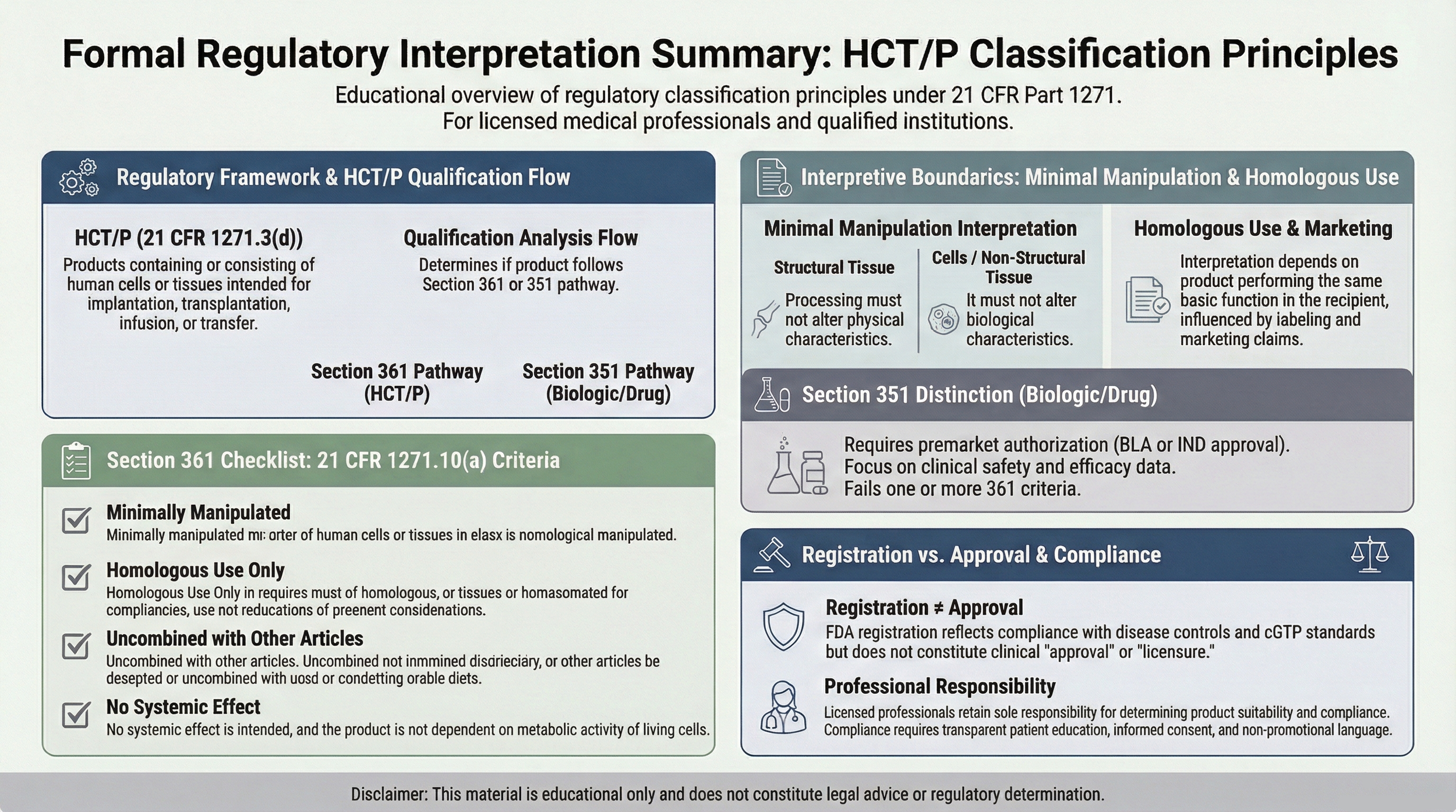
Regulatory Framework
Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 and Section 361 of the Public Health Service Act.
Products qualifying solely under Section 361 are subject primarily to communicable disease controls, establishment registration, donor eligibility requirements, and current good tissue practice (cGTP) standards.
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
Section 361 Qualification Criteria
For a product to qualify for regulation solely under Section 361, it must generally satisfy all applicable criteria, including:
- Minimal manipulation
- Homologous use only
- No impermissible combination with other articles
- No systemic effect or dependence on living cells’ metabolic activity, except as permitted under specific regulatory allowances
Failure to meet these criteria may result in regulation under Section 351 as a biological product, which may require additional regulatory pathways.
Minimal Manipulation & Homologous Use
Minimal manipulation standards differ depending on whether the tissue is structural or cellular in nature.
Homologous use requires that the product perform the same basic function in the recipient as it did in the donor.
Intended use, labeling, and surrounding communications may influence regulatory interpretation.
Non-Cellular & Structural Distinctions
Structural tissue materials, including extracellular matrix-derived products, are evaluated based on preservation of structural characteristics.
Products described as non-cellular formulations may require separate classification analysis depending on composition, processing, and intended context.
Compliance Communication Standards
Communications regarding tissue-based products should avoid representations that imply FDA approval for diagnosing, treating, curing, or preventing specific diseases.
Language used in professional and patient-facing contexts should remain consistent with homologous use limitations and regulatory classification.
Professional Responsibility
Licensed medical professionals and institutions are responsible for conducting independent regulatory evaluation and ensuring compliance with applicable federal and state requirements.
Nothing contained herein constitutes legal advice or regulatory determination.
Reference Material
For full regulatory text, refer to:
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR 1271. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

