Professional Regulatory FAQ — Licensed Medical Professionals
This regulatory FAQ provides structured clarification regarding HCT/P classification under 21 CFR Part 1271 Section 361, including Section 361 vs Section 351 distinctions, minimal manipulation standards, homologous use interpretation, donor eligibility requirements, and communicable disease control obligations.
1. What regulatory framework applies to certain biologic products?
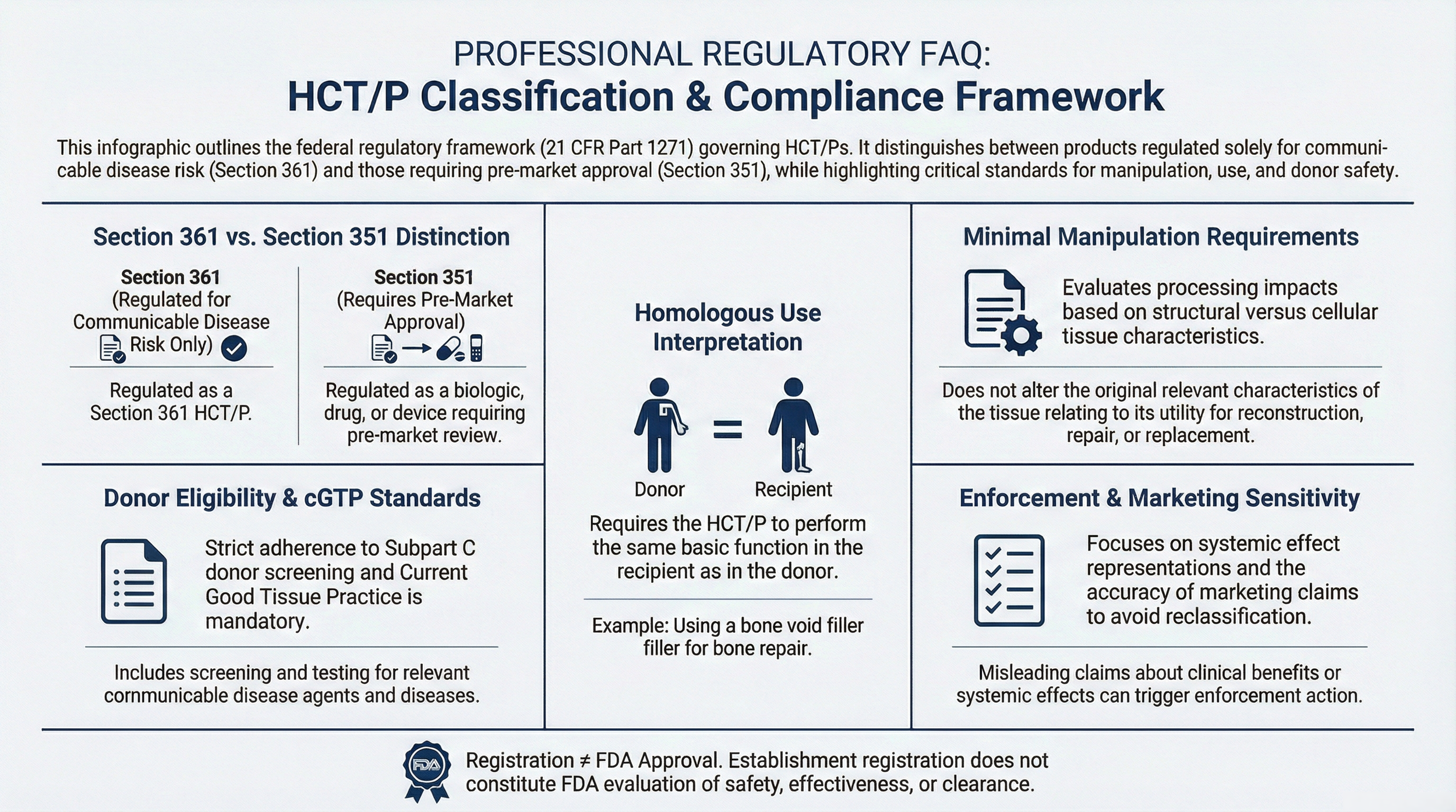
Certain products may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. If qualification criteria are met, the product may fall within the Section 361 HCT/P communicable disease control pathway. If criteria are not met, regulation may shift to Section 351 as a biological product.
2. What is the difference between Section 361 and Section 351?
Section 361 HCT/Ps are regulated primarily for communicable disease control. Section 351 products are regulated as biological products and may require premarket approval or biologics licensure. The 361 vs 351 distinction is central to regulatory risk evaluation.
3. What defines minimal manipulation under 21 CFR 1271?
The minimal manipulation standard differs depending on whether the tissue is structural or cellular. Structural tissues must retain original relevant structural characteristics. Cellular or non-structural tissues must retain relevant biological characteristics.
4. What is homologous use under 21 CFR 1271.3(c)?
Homologous use means the HCT/P performs the same basic function in the recipient as it did in the donor.
5. What are donor eligibility requirements?
Donor eligibility determination under 21 CFR 1271 Subpart C includes medical history screening, communicable disease risk evaluation, and laboratory testing.
Educational reference only. Certain products may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 Section 361. Registration does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.

