Medical Oversight & Clinical Advisory Disclosure
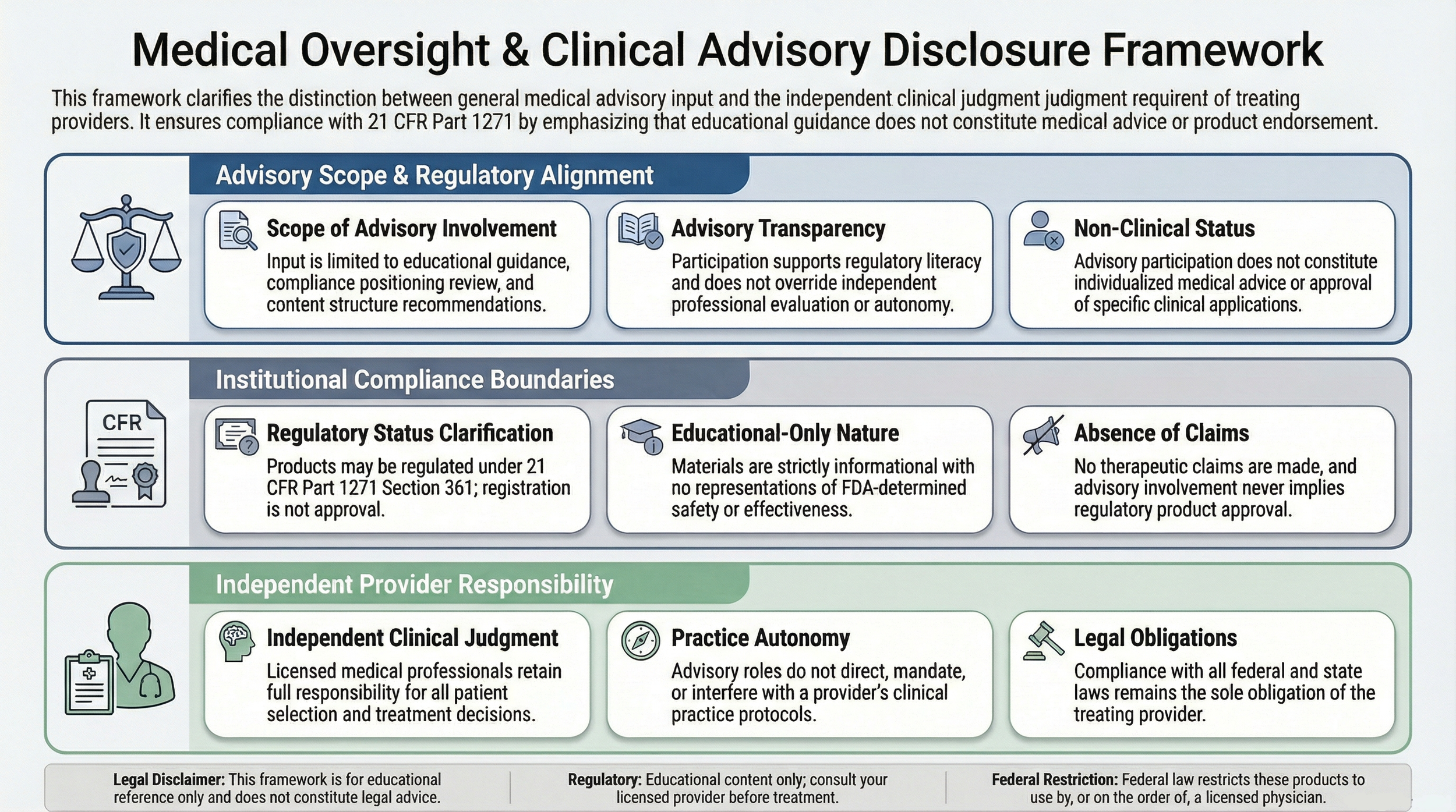
This disclosure outlines the scope and limitations of medical oversight and clinical advisory involvement in connection with educational materials presented on this platform.
It is intended to clarify professional roles, advisory boundaries, and provider responsibility consistent with applicable regulatory frameworks.
Scope of Medical Advisory Involvement
Clinical advisors or medical directors associated with referenced suppliers may provide input regarding general educational content, regulatory alignment, and compliance positioning.
Such involvement does not constitute individualized medical advice, product endorsement, or approval of specific clinical applications.
No FDA Approval or Evaluation
Products referenced may be regulated under 21 CFR Part 1271 and Section 361 of the Public Health Service Act.
Registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness.
Medical oversight participation does not imply regulatory approval.
Independent Clinical Judgment
Licensed medical professionals are solely responsible for exercising independent clinical judgment in evaluating patient eligibility, procedural considerations, and applicable regulatory requirements.
No content on this platform should be interpreted as directing, prescribing, or mandating clinical decisions.
Educational Nature of Content
All materials are provided for educational and informational reference only. Content is structured to reflect regulatory classifications and homologous use limitations consistent with 21 CFR 1271.3 .
No representations are made that products are approved to diagnose, treat, cure, or prevent any disease.
Conflict of Interest & Transparency
Where advisory relationships exist between suppliers and clinical professionals, such relationships are structured to support regulatory literacy and compliance alignment.
Advisory roles do not override independent provider responsibility or applicable federal and state laws.
Professional Responsibility
Licensed medical professionals and institutions are responsible for conducting independent regulatory evaluation and ensuring compliance with applicable federal and state requirements.
Nothing contained herein constitutes legal advice, regulatory determination, or individualized medical guidance.
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271 . These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

