Formal Regulatory Interpretation Summary
This document provides a structured educational summary of regulatory interpretation principles applicable to Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271.
It is intended for licensed medical professionals and qualified institutions evaluating regulatory classification boundaries.
Regulatory Framework Overview
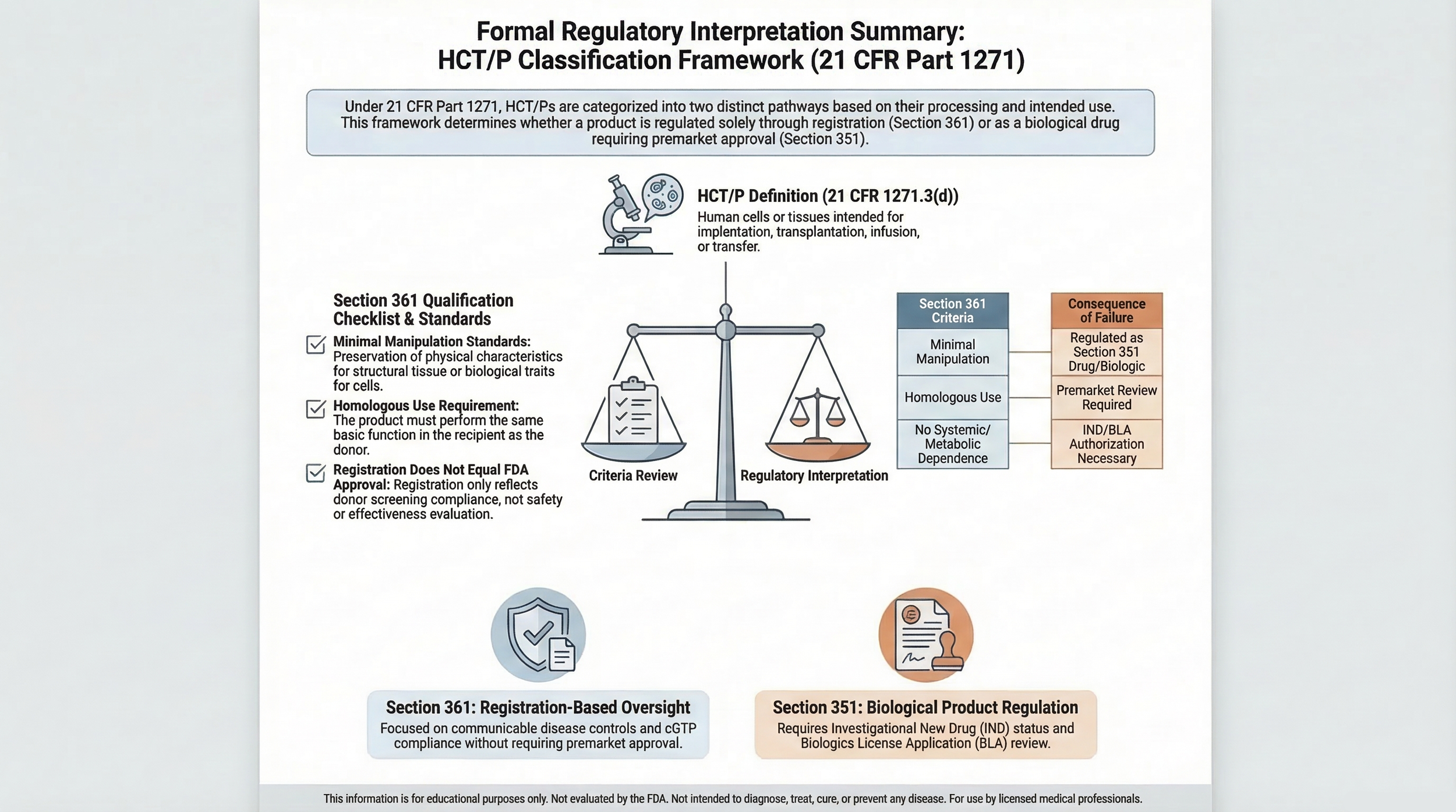
HCT/Ps are defined in 21 CFR 1271.3(d) as articles containing or consisting of human cells or tissues intended for implantation, transplantation, infusion, or transfer into a human recipient.
Regulatory oversight depends on whether a product qualifies solely under Section 361 of the Public Health Service Act or requires regulation as a biological product under Section 351.
Section 361 Qualification Criteria
To qualify solely under Section 361, a product must generally meet all applicable criteria:
- Minimal manipulation
- Homologous use only
- No combination with other articles (with limited exceptions)
- No systemic effect or dependence on living cells’ metabolic activity, except where permitted
Failure to satisfy any criterion may result in regulation under Section 351.
Minimal Manipulation: Structural vs Cellular Tissue
For structural tissue, minimal manipulation means that processing does not alter the original relevant characteristics relating to reconstruction, repair, or replacement.
For cells or non-structural tissues, minimal manipulation refers to processing that does not alter relevant biological characteristics.
Homologous Use Interpretation
Homologous use requires that the product perform the same basic function in the recipient as in the donor.
Intended use statements, labeling, and marketing communications may influence regulatory interpretation of homologous use.
Section 351 Distinction
Products that do not meet Section 361 criteria may be regulated under Section 351 as biological products, potentially requiring Investigational New Drug (IND) applications or Biologics License Applications (BLA).
Registration vs Approval
Establishment registration under 21 CFR Part 1271 does not constitute FDA approval or evaluation of safety or effectiveness.
Professional Responsibility
Licensed medical professionals retain responsibility for independent regulatory evaluation and compliance with federal and state requirements.
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

