FDA Guidance — Minimal Manipulation (21 CFR Part 1271)
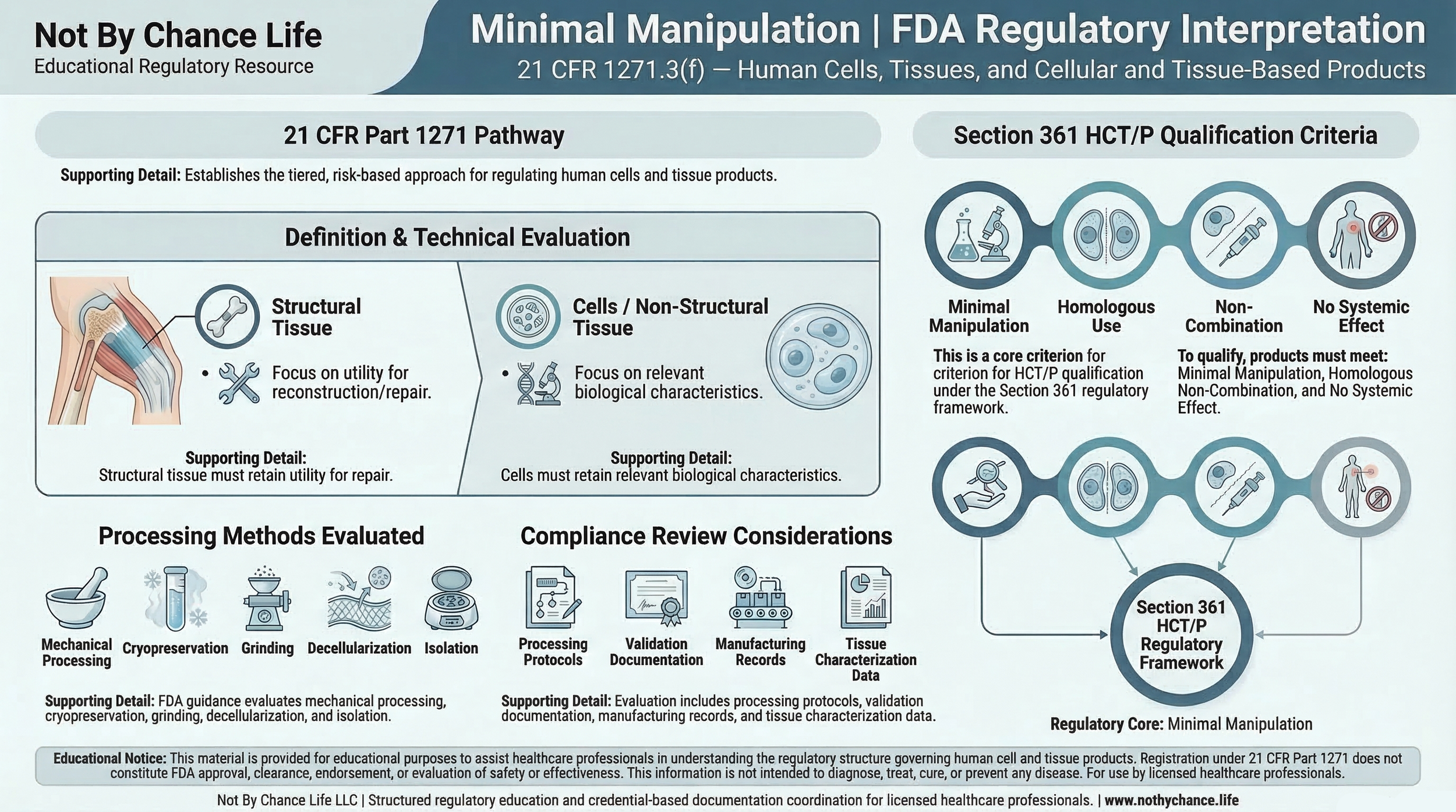
The concept of minimal manipulation is a central regulatory criterion used to determine whether a human cell or tissue product may qualify for regulation solely under Section 361 of the Public Health Service Act and 21 CFR Part 1271.
The U.S. Food and Drug Administration (FDA) has issued guidance documents interpreting how minimal manipulation standards apply to tissues and cellular materials regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).
These guidance materials help manufacturers, tissue establishments, compliance professionals, and healthcare institutions understand how processing steps may affect regulatory classification.
Regulatory Definition of Minimal Manipulation
Minimal manipulation is defined in 21 CFR 1271.3(f). The definition is applied differently depending on whether the product is categorized as structural tissue or cells / non-structural tissue.
Processing must not alter the original relevant characteristics of the tissue relating to its utility for reconstruction, repair, or replacement.
Cells or Non-Structural TissueProcessing must not alter the relevant biological characteristics of the cells or tissues.
Further discussion of these distinctions is provided in the page addressing structural tissue versus cellular tissue classification .
Role of FDA Guidance Documents
FDA guidance documents provide interpretive clarification regarding how minimal manipulation standards are applied during regulatory review. These documents outline regulatory reasoning and provide examples of processing methods that may alter relevant tissue characteristics.
Examples frequently discussed within FDA guidance include:
- Mechanical processing or shaping of structural tissue
- Decellularization methods
- Grinding or sizing procedures
- Cryopreservation and storage techniques
- Isolation, expansion, or culture of cellular components
Interpretation of minimal manipulation is typically evaluated alongside homologous use requirements and other criteria outlined in 21 CFR 1271.10(a).
Relationship to Section 361 Regulatory Qualification
Minimal manipulation is one of four regulatory criteria used to determine whether a product may qualify for regulation under Section 361 rather than the biologics licensing pathway defined under Section 351.
Additional regulatory criteria include:
- Homologous use
- Non-combination with other active agents
- Limited systemic effect or metabolic activity
These regulatory distinctions are discussed further in the overview of Section 361 versus Section 351 classification .
Importance for Regulatory Compliance
Manufacturers, tissue establishments, and healthcare institutions reviewing biologic materials commonly examine processing documentation to determine whether tissue characteristics remain consistent with minimal manipulation standards.
Regulatory review may include examination of:
- Processing and manufacturing protocols
- Laboratory validation documentation
- Manufacturing batch records
- Tissue characterization and analytical data
These evaluations typically occur alongside review of donor eligibility determination requirements and current good tissue practice (cGTP) safeguards .
Educational Purpose of This Resource
This page provides educational information summarizing FDA guidance related to minimal manipulation standards within the regulatory framework governing Human Cells, Tissues, and Cellular and Tissue-Based Products.
Additional regulatory education materials are available within the Regulatory Resource Library .
Related Regulatory Topics
- 21 CFR Part 1271 Overview
- Minimal Manipulation Explained
- Homologous Use Interpretation
- Structural Tissue vs Cellular Tissue
- Regulatory Resource Library
Educational reference only. Certain referenced materials may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR Part 1271. Establishment registration under 21 CFR Part 1271 does not constitute FDA approval, clearance, or evaluation of safety or effectiveness. This information is not intended to diagnose, treat, cure, or prevent any disease.

