Exosomes and Regulatory Status
Exosomes are small extracellular vesicles released by cells that participate in cell-to-cell communication and signaling processes. These vesicles contain proteins, lipids, and nucleic acids that can influence biological activity.
Within regenerative medicine discussions, exosomes are sometimes referenced alongside other biologic materials. However, their regulatory classification is distinct from structural tissue products regulated under the Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/P) framework.
Understanding the FDA regulatory status of exosome products is important for healthcare professionals and regulatory reviewers evaluating biologic materials in the United States.
The infographic below summarizes how exosome products are commonly interpreted within the FDA regulatory framework compared with structural tissue products regulated under 21 CFR Part 1271.
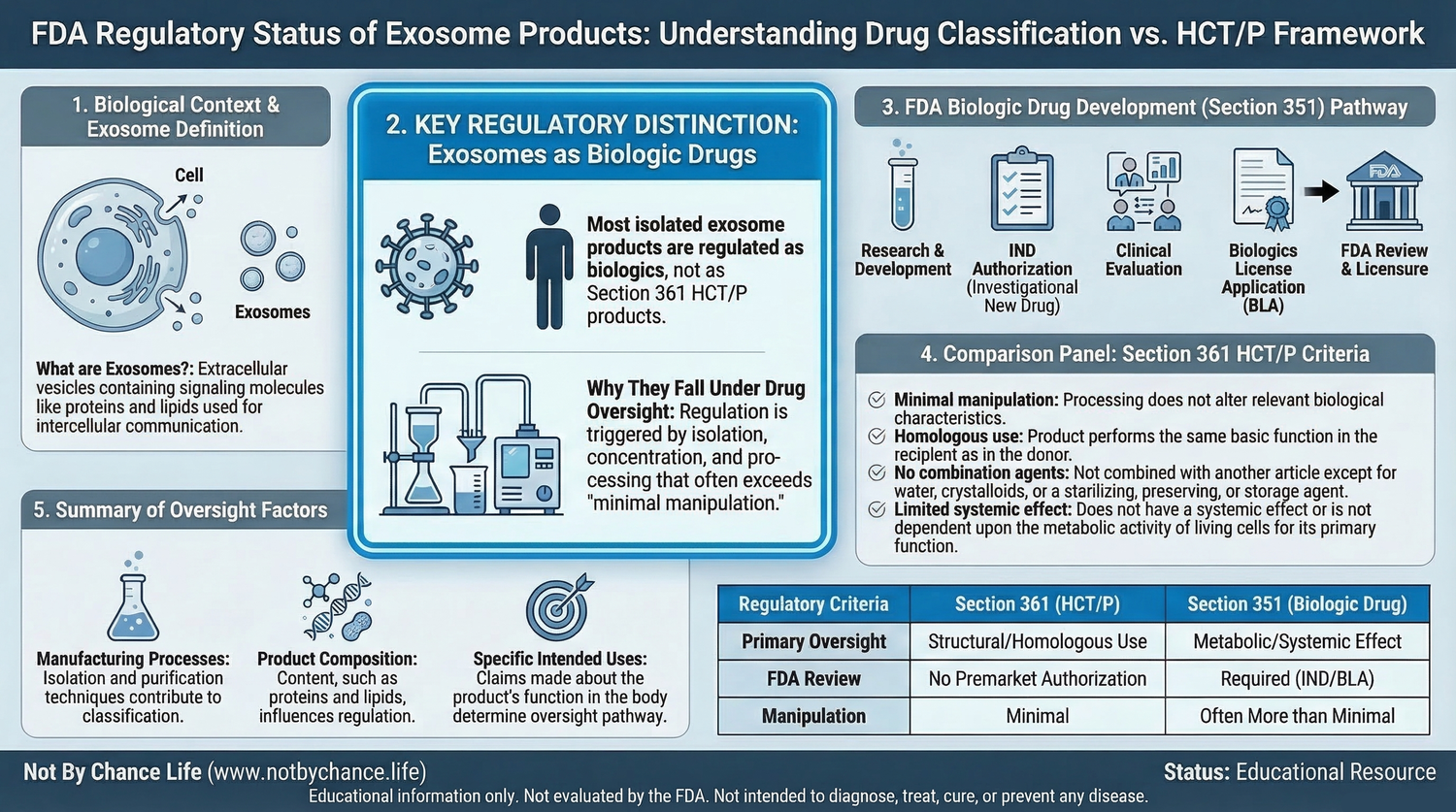
Regulatory Classification
Current regulatory interpretation generally classifies isolated or manufactured exosome products as biologic drugs rather than HCT/Ps regulated solely under 21 CFR Part 1271.
- Exosome products are generally considered biologic drugs
- Drug products require formal regulatory pathways
- This includes Investigational New Drug (IND) authorization or Biologics License Application (BLA)
Because exosomes are typically isolated, concentrated, or otherwise processed from their source material, they may fall outside the regulatory definitions required for classification as a Section 361 HCT/P.
As a result, commercial distribution or clinical use of exosome products generally requires regulatory oversight consistent with drug development frameworks.
Distinction from Structural Tissue Products
Structural human tissue products regulated under Section 361 must meet several criteria, including:
- Minimal manipulation
- Homologous use
- No combination with other active agents
- No systemic effect unless specific exceptions apply
Isolated extracellular vesicles such as exosomes generally do not meet these criteria when processed or marketed independently.
Educational Context
Educational discussions of extracellular vesicles are common in scientific literature related to cellular signaling and tissue biology. However, the presence of research publications does not indicate regulatory approval for clinical use.
Regulatory classification depends on manufacturing processes, product composition, intended use, and compliance with applicable federal regulations.
Professional Evaluation
Healthcare professionals evaluating biologic materials should review regulatory documentation, manufacturing standards, and available compliance information before considering the use of any biologic product.
Transparent documentation helps ensure that materials used in clinical settings are aligned with applicable regulatory frameworks and professional standards.
References
- FDA Public Safety Notification on Exosome Products
- FDA Guidance for Industry: Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps)
- 21 CFR Part 1271 – Human Cells, Tissues, and Cellular and Tissue-Based Products
- Scientific literature on extracellular vesicles and intercellular signaling
This information is provided for educational purposes only.
These materials have not been evaluated by the Food and Drug Administration.
Nothing on this page is intended to diagnose, treat, cure, or prevent any disease.
Information is intended for licensed healthcare professionals and qualified industry participants.

