Clinical Evidence & Research Overview
This page provides an educational overview of clinical evidence considerations related to tissue-based and biologic product categories discussed within the HCT/P regulatory framework.
It is intended for licensed medical professionals and qualified institutions evaluating available literature, case data, and research context.
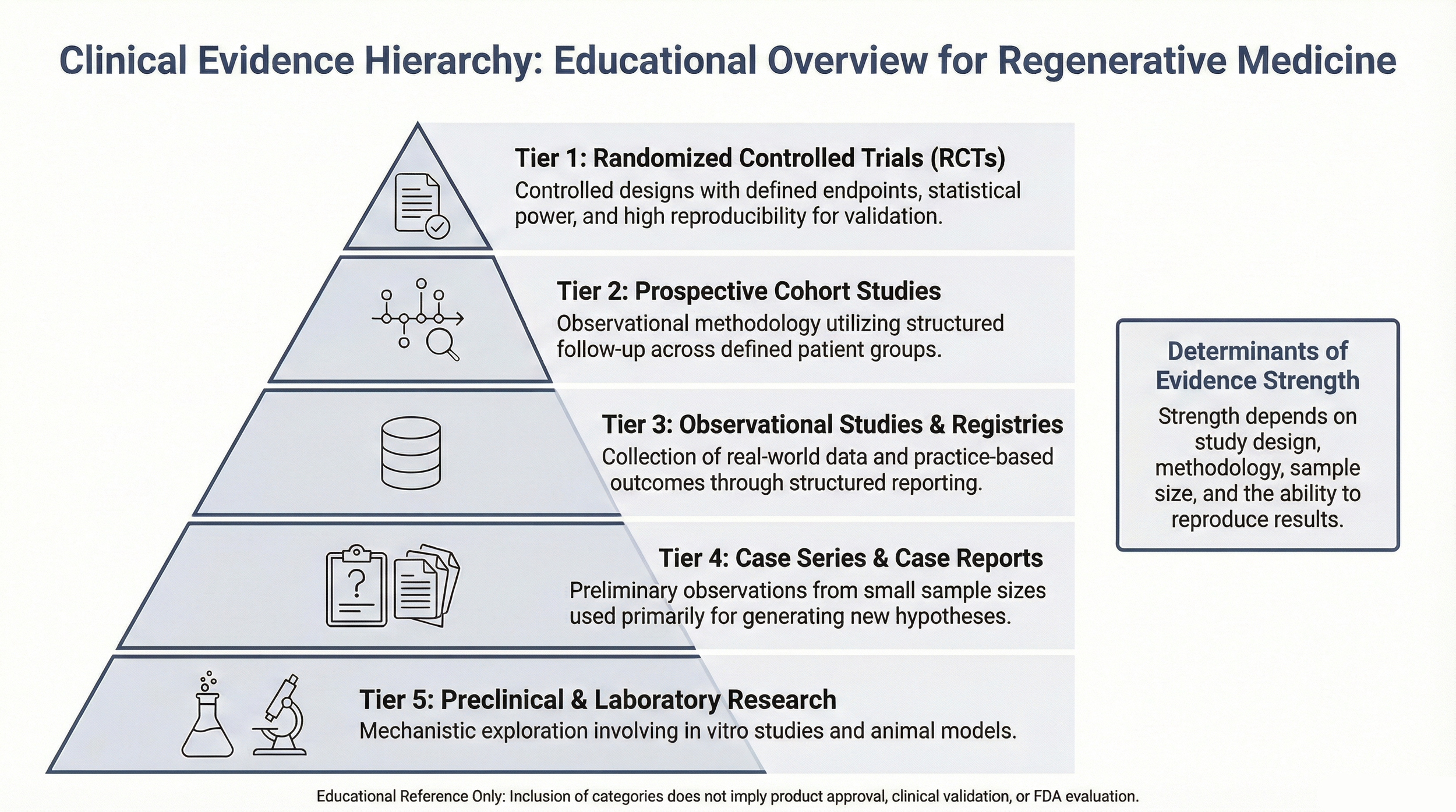
Evidence Hierarchy in Regenerative Medicine
Clinical evidence in regenerative medicine may range from preclinical laboratory research to observational studies, case reports, prospective registries, and randomized controlled trials.
The strength of evidence depends on study design, sample size, controls, statistical methodology, and reproducibility.
Case Reports & Observational Data
Case reports and small observational studies may provide preliminary insight into product tolerability and short-term clinical observations.
Such reports are not equivalent to randomized controlled trials and should be interpreted within appropriate methodological limits.
Safety Monitoring Considerations
Safety evaluation in tissue-based product categories may include monitoring for adverse events, local reactions, and procedural complications consistent with standard clinical practice.
Regulatory registration under 21 CFR Part 1271 does not imply FDA evaluation of safety or effectiveness.
Research vs Approved Indications
Products regulated solely under Section 361 are not subject to premarket approval pathways required for Section 351 biological products.
Providers should distinguish between investigational research, emerging literature, and FDA-approved indications when discussing tissue-based products.
Professional Evaluation & Due Diligence
Licensed professionals are responsible for reviewing available literature, assessing methodological rigor, and applying independent clinical judgment consistent with regulatory boundaries.
Product communications should remain consistent with homologous use limitations and avoid representations that imply approval for diagnosing, treating, curing, or preventing specific diseases.
Ongoing Data Collection & Registries
Real-world data collection, patient registries, and structured follow-up systems may contribute to broader understanding of safety and clinical observations over time.
Such data should be interpreted within appropriate research and methodological frameworks.
Reference Materials
For regulatory reference, consult:
Educational reference only. Certain products referenced may be regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under 21 CFR 1271. These materials have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

